|
51
|
Erisik D, Ozdil B, Acikgoz E, Asker Abdikan CS, Yesin TK, Aktug H. Differences and Similarities between Colorectal Cancer Cells and Colorectal Cancer Stem Cells: Molecular Insights and Implications. ACS OMEGA 2023; 8:30145-30157. [PMID: 37636966 PMCID: PMC10448492 DOI: 10.1021/acsomega.3c02681] [Citation(s) in RCA: 1] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Figures] [Subscribe] [Scholar Register] [Received: 04/19/2023] [Accepted: 07/26/2023] [Indexed: 08/29/2023]
Abstract
Malignant tumors are formed by diverse groups of cancer cells. Cancer stem cells (CSCs) are a subpopulation of heterogeneous cells identified in tumors that have the ability to self-renew and differentiate. Colorectal cancer (CRC), the third most frequent malignant tumor, is progressively being supported by evidence suggesting that CSCs are crucial in cancer development. We aim to identify molecular differences between CRC cells and CRC CSCs, as well as the effects of those differences on cell behavior in terms of migration, EMT, pluripotency, morphology, cell cycle/control, and epigenetic characteristics. The HT-29 cell line (human colorectal adenocarcinoma) and HT-29 CSCs (HT-29 CD133+/CD44+ cells) were cultured for 72 h. The levels of E-cadherin, KLF4, p53, p21, p16, cyclin D2, HDAC9, and P300 protein expression were determined using immunohistochemistry staining. The migration of cells was assessed by employing the scratch assay technique. Additionally, the scanning electron microscopy method was used to examine the morphological features of the cells, and their peripheral/central elemental ratios were compared with the help of EDS. Furthermore, a Muse cell cycle kit was utilized to determine the cell cycle analysis. The HT-29 CSC group exhibited high levels of expression for E-cadherin, p53, p21, p16, cyclin D2, HDAC9, and P300, whereas KLF4 was found to be high in the HT-29. The two groups did not exhibit any statistically significant differences in the percentages of cell cycle phases. The identification of specific CSC characteristics will allow for earlier cancer detection and the development of more effective precision oncology options.
Collapse
Affiliation(s)
- Derya Erisik
- Department
of Histology and Embryology, Faculty of Medicine, Ege University, Izmir 35100, Turkey
| | - Berrin Ozdil
- Department
of Histology and Embryology, Faculty of Medicine, Ege University, Izmir 35100, Turkey
- Department
of Histology and Embryology, Faculty of Medicine, Suleyman Demirel University, Isparta 32260, Turkey
| | - Eda Acikgoz
- Department
of Histology and Embryology, Faculty of Medicine, Yuzuncu Yil University, Van 65080, Turkey
| | | | - Taha Kadir Yesin
- Department
of Histology and Embryology, Faculty of Medicine, Ege University, Izmir 35100, Turkey
| | - Huseyin Aktug
- Department
of Histology and Embryology, Faculty of Medicine, Ege University, Izmir 35100, Turkey
| |
Collapse
|
|
52
|
He Z, Fan F, Xu Z, Zhang M, Zhao R, Ke X, Wang Q, Yan S, Zheng H. Downregulation of CLDN6 inhibits cell migration and invasion and promotes apoptosis by regulation of the JAK2/STAT3 signaling pathway in hepatocellular carcinoma. Transl Cancer Res 2023; 12:1753-1764. [PMID: 37588735 PMCID: PMC10425638 DOI: 10.21037/tcr-23-19] [Citation(s) in RCA: 1] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/05/2023] [Accepted: 06/07/2023] [Indexed: 08/18/2023]
Abstract
Background High expression of CLDN6 in hepatocellular carcinoma (HCC) has been widely reported. During this research, CLDN6's effect on the infiltration, migration, and apoptosis of HCC cells was investigated. Methods Initially, the knockdown and overexpression of CLDN6 in HCC cells were carried out by short interfering RNA (siRNA) and plasmid transfection. The transfection efficiency was detected by means of a quantitative real-time polymerase chain reaction (qRT-PCR) assay, immunofluorescence staining, and Western blot analysis. Transwell and wound-healing assays were employed for the detection of invasion and migration ability. CCK-8 assay and flow cytometry were utilized for the detection of apoptosis. Finally, analysis of the expression of pathway-related proteins (JAK2, STAT3, p-JAK2, and p-STAT3) and the regulation of apoptotic responses (by measurement of cleaved caspase-3, Bax, and Bcl-2 levels) was carried out. Results When CLDN6 was knocked down, the cellular invasion and migration ability decreased, and apoptosis increased, which decreased p-JAK2, p-STAT3, and anti-apoptotic protein bcl-2 expression. Furthermore, an elevation was observed in cleaved caspase-3 and Bax expression levels. Contrarily, upon overexpression of CDLN6, the aforementioned experimental results were reversed. Conclusions CLDN6 knockdown results in the inhibition of HCC cells' infiltration and migration and promotes apoptosis via downregulation of the JAK2/STAT3 signaling pathway.
Collapse
Affiliation(s)
- Zhiyi He
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Fangtian Fan
- Faculty of Pharmacy, Bengbu Medical College, Bengbu, China
| | - Zhidong Xu
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Mei Zhang
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Rui Zhao
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Xiquan Ke
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Qizhi Wang
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Shanjun Yan
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| | - Hailun Zheng
- Department of Gastroenterology, The First Affiliated Hospital of Bengbu Medical College, Bengbu, China
| |
Collapse
|
|
53
|
Jerin S, Harvey AJ, Lewis A. Therapeutic Potential of Protein Tyrosine Kinase 6 in Colorectal Cancer. Cancers (Basel) 2023; 15:3703. [PMID: 37509364 PMCID: PMC10377740 DOI: 10.3390/cancers15143703] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/24/2023] [Revised: 07/10/2023] [Accepted: 07/19/2023] [Indexed: 07/30/2023] Open
Abstract
PTK6, a non-receptor tyrosine kinase, modulates the pathogenesis of breast and prostate cancers and is recognized as a biomarker of breast cancer prognosis. There are over 30 known substrates of PTK6, including signal transducers, transcription factors, and RNA-binding proteins. Many of these substrates are known drivers of other cancer types, such as colorectal cancer. Colon and rectal tumors also express higher levels of PTK6 than the normal intestine suggesting a potential role in tumorigenesis. However, the importance of PTK6 in colorectal cancer remains unclear. PTK6 inhibitors such as XMU-MP-2 and Tilfrinib have demonstrated potency and selectivity in breast cancer cells when used in combination with chemotherapy, indicating the potential for PTK6 targeted therapy in cancer. However, most of these inhibitors are yet to be tested in other cancer types. Here, we discuss the current understanding of the function of PTK6 in normal intestinal cells compared with colorectal cancer cells. We review existing PTK6 targeting therapeutics and explore the possibility of PTK6 inhibitory therapy for colorectal cancer.
Collapse
Affiliation(s)
- Samanta Jerin
- Centre for Genome Engineering and Maintenance, Division of Biosciences, Department of Life Sciences, College of Health and Life Sciences, Brunel University London, Uxbridge UB8 3PH, UK
| | - Amanda J Harvey
- Centre for Genome Engineering and Maintenance, Institute for Health Medicine and Environments, Brunel University London, Uxbridge UB8 3PH, UK
| | - Annabelle Lewis
- Centre for Genome Engineering and Maintenance, Division of Biosciences, Department of Life Sciences, College of Health and Life Sciences, Brunel University London, Uxbridge UB8 3PH, UK
| |
Collapse
|
|
54
|
Shi W, Chen J, Yao N, Wu T, Suo X, Wang Q, Liu J, Yu G, Zhang K. The prognostic ability of radiotherapy of different colorectal cancer histological subtypes and tumor sites. Sci Rep 2023; 13:11758. [PMID: 37474552 PMCID: PMC10359278 DOI: 10.1038/s41598-023-38853-9] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 02/16/2023] [Accepted: 07/16/2023] [Indexed: 07/22/2023] Open
Abstract
The prognostic significance of radiotherapy (RT) for colorectal cancer (CRC) has shown conflicting results, particularly among different pathological subtypes, including adenocarcinoma (AC), mucinous adenocarcinoma (MC), and signet-ring cell carcinoma (SR). This study analyzed the prognosis of three pathological CRC types and focused on the prognostic significance of RT on three CRC histological subtypes. Patients diagnosed with AC (n = 54,174), MC (n = 3813), and SR (n = 664) in the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) database (2010-2017) were evaluated. Cox regression models and competitive risk models were built to assess the effect of RT on the risk of CRC-associated death. Potential interactions between RT and stratified variables including age, sex, and tumor location were examined by multiplicative models. Compared with AC patients, SR patients had the worst overall survival (OS) among 3 subtypes of CRC (log-rank test, p < 0.001). Compared with patients who did not receive radiotherapy, RT was associated with a 1.09-fold (HR = 1.09, 95%[CI]: 1.03, 1.15) elevated risk of death among AC patients. In the SR group, RT significantly reduced the risk of death by 39% (HR = 0.61, 95%[CI]: 0.39-0.95). However, RT did not appear to independently influence survival in the MC group (HR = 0.96, 95%[CI]: 0.77, 1.21). In the subgroup analysis, tumor location (colon and rectum) significantly modified the association between RT and the risk of death among the AC and SR patients (p for interaction < 0.05). SR patients exhibited a worse OS (overall survival) than AC patients, and the effect of RT varied according to CRC histological subtypes. This can ultimately lead to more personalized and effective treatment strategies for CRC patients.
Collapse
Affiliation(s)
- Wenzai Shi
- Department of Hepatobiliary Surgery, Peking University International Hospital, Life Park Road No.1 Life Science Park of Zhong Guancun, Chang Ping District, Beijing, 102206, China
| | - Jianfei Chen
- Department of Surgery, Beijing Shijitan Hospital, Capital Medical University, Beijing, 100038, China
- Ninth School of Clinical Medicine, Peking University, Beijing, 100038, China
- School of Oncology, Capital Medical University, Beijing, 100038, China
| | - Nan Yao
- Department of General Surgery, Aerospace Center Hospital, Beijing, 100089, China
| | - Tiantian Wu
- Department of Hepatobiliary Surgery, Peking University International Hospital, Life Park Road No.1 Life Science Park of Zhong Guancun, Chang Ping District, Beijing, 102206, China
| | - Xiaopeng Suo
- Department of Hepatobiliary Surgery, Peking University International Hospital, Life Park Road No.1 Life Science Park of Zhong Guancun, Chang Ping District, Beijing, 102206, China
| | - Qiang Wang
- Department of Hepatobiliary Surgery, Peking University International Hospital, Life Park Road No.1 Life Science Park of Zhong Guancun, Chang Ping District, Beijing, 102206, China
| | - Jun Liu
- Department of Hepatobiliary Surgery, Peking University International Hospital, Life Park Road No.1 Life Science Park of Zhong Guancun, Chang Ping District, Beijing, 102206, China
| | - Guoyong Yu
- Department of Nephrology, Beijing University of Chinese Medicine Affiliated Dongzhimen Hospital, East 4th North Street 279, Dongcheng District, Beijing, 100007, China.
| | - Keming Zhang
- Department of Hepatobiliary Surgery, Peking University International Hospital, Life Park Road No.1 Life Science Park of Zhong Guancun, Chang Ping District, Beijing, 102206, China.
| |
Collapse
|
|
55
|
Averbeck D. Low-Dose Non-Targeted Effects and Mitochondrial Control. Int J Mol Sci 2023; 24:11460. [PMID: 37511215 PMCID: PMC10380638 DOI: 10.3390/ijms241411460] [Citation(s) in RCA: 6] [Impact Index Per Article: 6.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/14/2023] [Revised: 06/26/2023] [Accepted: 06/29/2023] [Indexed: 07/30/2023] Open
Abstract
Non-targeted effects (NTE) have been generally regarded as a low-dose ionizing radiation (IR) phenomenon. Recently, regarding long distant abscopal effects have also been observed at high doses of IR) relevant to antitumor radiation therapy. IR is inducing NTE involving intracellular and extracellular signaling, which may lead to short-ranging bystander effects and distant long-ranging extracellular signaling abscopal effects. Internal and "spontaneous" cellular stress is mostly due to metabolic oxidative stress involving mitochondrial energy production (ATP) through oxidative phosphorylation and/or anaerobic pathways accompanied by the leakage of O2- and other radicals from mitochondria during normal or increased cellular energy requirements or to mitochondrial dysfunction. Among external stressors, ionizing radiation (IR) has been shown to very rapidly perturb mitochondrial functions, leading to increased energy supply demands and to ROS/NOS production. Depending on the dose, this affects all types of cell constituents, including DNA, RNA, amino acids, proteins, and membranes, perturbing normal inner cell organization and function, and forcing cells to reorganize the intracellular metabolism and the network of organelles. The reorganization implies intracellular cytoplasmic-nuclear shuttling of important proteins, activation of autophagy, and mitophagy, as well as induction of cell cycle arrest, DNA repair, apoptosis, and senescence. It also includes reprogramming of mitochondrial metabolism as well as genetic and epigenetic control of the expression of genes and proteins in order to ensure cell and tissue survival. At low doses of IR, directly irradiated cells may already exert non-targeted effects (NTE) involving the release of molecular mediators, such as radicals, cytokines, DNA fragments, small RNAs, and proteins (sometimes in the form of extracellular vehicles or exosomes), which can induce damage of unirradiated neighboring bystander or distant (abscopal) cells as well as immune responses. Such non-targeted effects (NTE) are contributing to low-dose phenomena, such as hormesis, adaptive responses, low-dose hypersensitivity, and genomic instability, and they are also promoting suppression and/or activation of immune cells. All of these are parts of the main defense systems of cells and tissues, including IR-induced innate and adaptive immune responses. The present review is focused on the prominent role of mitochondria in these processes, which are determinants of cell survival and anti-tumor RT.
Collapse
Affiliation(s)
- Dietrich Averbeck
- Laboratory of Cellular and Molecular Radiobiology, PRISME, UMR CNRS 5822/IN2P3, IP2I, Lyon-Sud Medical School, University Lyon 1, 69921 Oullins, France
| |
Collapse
|
|
56
|
Ponomarev AS, Gilazieva ZE, Solovyova VV, Rizvanov AA. Molecular Mechanisms of Tumor Cell Stemness Modulation during Formation of Spheroids. BIOCHEMISTRY. BIOKHIMIIA 2023; 88:979-994. [PMID: 37751868 DOI: 10.1134/s0006297923070106] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Subscribe] [Scholar Register] [Received: 01/31/2023] [Revised: 05/30/2023] [Accepted: 06/07/2023] [Indexed: 09/28/2023]
Abstract
Cancer stem cells (CSCs), their properties and interaction with microenvironment are of interest in modern medicine and biology. There are many studies on the emergence of CSCs and their involvement in tumor pathogenesis. The most important property inherent to CSCs is their stemness. Stemness combines ability of the cell to maintain its pluripotency, give rise to differentiated cells, and interact with environment to maintain a balance between dormancy, proliferation, and regeneration. While adult stem cells exhibit these properties by participating in tissue homeostasis, CSCs behave as their malignant equivalents. High tumor resistance to therapy, ability to differentiate, activate angiogenesis and metastasis arise precisely due to the stemness of CSCs. These cells can be used as a target for therapy of different types of cancer. Laboratory models are needed to study cancer biology and find new therapeutic strategies. A promising direction is three-dimensional tumor models or spheroids. Such models exhibit properties resembling stemness in a natural tumor. By modifying spheroids, it becomes possible to investigate the effect of therapy on CSCs, thus contributing to the development of anti-tumor drug test systems. The review examines the niche of CSCs, the possibility of their study using three-dimensional spheroids, and existing markers for assessing stemness of CSCs.
Collapse
Affiliation(s)
- Aleksei S Ponomarev
- Kazan (Volga Region) Federal University, Kazan, Republic of Tatarstan, 420008, Russia
| | - Zarema E Gilazieva
- Kazan (Volga Region) Federal University, Kazan, Republic of Tatarstan, 420008, Russia
| | - Valeriya V Solovyova
- Kazan (Volga Region) Federal University, Kazan, Republic of Tatarstan, 420008, Russia
| | - Albert A Rizvanov
- Kazan (Volga Region) Federal University, Kazan, Republic of Tatarstan, 420008, Russia.
| |
Collapse
|
|
57
|
Entrialgo-Cadierno R, Cueto-Ureña C, Welch C, Feliu I, Macaya I, Vera L, Morales X, Michelina SV, Scaparone P, Lopez I, Darbo E, Erice O, Vallejo A, Moreno H, Goñi-Salaverri A, Lara-Astiaso D, Halberg N, Cortes-Dominguez I, Guruceaga E, Ambrogio C, Lecanda F, Vicent S. The phospholipid transporter PITPNC1 links KRAS to MYC to prevent autophagy in lung and pancreatic cancer. Mol Cancer 2023; 22:86. [PMID: 37210549 DOI: 10.1186/s12943-023-01788-w] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/18/2022] [Accepted: 05/11/2023] [Indexed: 05/22/2023] Open
Abstract
BACKGROUND The discovery of functionally relevant KRAS effectors in lung and pancreatic ductal adenocarcinoma (LUAD and PDAC) may yield novel molecular targets or mechanisms amenable to inhibition strategies. Phospholipids availability has been appreciated as a mechanism to modulate KRAS oncogenic potential. Thus, phospholipid transporters may play a functional role in KRAS-driven oncogenesis. Here, we identified and systematically studied the phospholipid transporter PITPNC1 and its controlled network in LUAD and PDAC. METHODS Genetic modulation of KRAS expression as well as pharmacological inhibition of canonical effectors was completed. PITPNC1 genetic depletion was performed in in vitro and in vivo LUAD and PDAC models. PITPNC1-deficient cells were RNA sequenced, and Gene Ontology and enrichment analyses were applied to the output data. Protein-based biochemical and subcellular localization assays were run to investigate PITPNC1-regulated pathways. A drug repurposing approach was used to predict surrogate PITPNC1 inhibitors that were tested in combination with KRASG12C inhibitors in 2D, 3D, and in vivo models. RESULTS PITPNC1 was increased in human LUAD and PDAC, and associated with poor patients' survival. PITPNC1 was regulated by KRAS through MEK1/2 and JNK1/2. Functional experiments showed PITPNC1 requirement for cell proliferation, cell cycle progression and tumour growth. Furthermore, PITPNC1 overexpression enhanced lung colonization and liver metastasis. PITPNC1 regulated a transcriptional signature which highly overlapped with that of KRAS, and controlled mTOR localization via enhanced MYC protein stability to prevent autophagy. JAK2 inhibitors were predicted as putative PITPNC1 inhibitors with antiproliferative effect and their combination with KRASG12C inhibitors elicited a substantial anti-tumour effect in LUAD and PDAC. CONCLUSIONS Our data highlight the functional and clinical relevance of PITPNC1 in LUAD and PDAC. Moreover, PITPNC1 constitutes a new mechanism linking KRAS to MYC, and controls a druggable transcriptional network for combinatorial treatments.
Collapse
Affiliation(s)
- Rodrigo Entrialgo-Cadierno
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Cristina Cueto-Ureña
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Connor Welch
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
- Centro de Investigación Biomédica en Red de Cáncer (CIBERONC), Madrid, Spain
| | - Iker Feliu
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Irati Macaya
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Laura Vera
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Xabier Morales
- Imaging Unit and Cancer Imaging Laboratory, University of Navarra, CIMA, Pamplona, Spain
| | - Sandra Vietti Michelina
- Department of Molecular Biotechnology and Health Sciences, Molecular Biotechnology Centre, University of Torino, Turin, Italy
| | - Pietro Scaparone
- Department of Molecular Biotechnology and Health Sciences, Molecular Biotechnology Centre, University of Torino, Turin, Italy
| | - Ines Lopez
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Elodie Darbo
- University of Bordeaux, INSERM, BRIC, U 1312, F-33000, Bordeaux, France
| | - Oihane Erice
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Adrian Vallejo
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | - Haritz Moreno
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
| | | | - David Lara-Astiaso
- Molecular Therapies Program, University of Navarra, CIMA, Pamplona, Spain
- Wellcome - MRC Cambridge Stem Cell Institute (CSCI), Cambridge, UK
| | - Nils Halberg
- Department of Biomedicine, University of Bergen, Bergen, Norway
| | - Ivan Cortes-Dominguez
- Imaging Unit and Cancer Imaging Laboratory, University of Navarra, CIMA, Pamplona, Spain
- Bioinformatics Platform, University of Navarra, CIMA, Pamplona, Spain
| | - Elizabeth Guruceaga
- Bioinformatics Platform, University of Navarra, CIMA, Pamplona, Spain
- IdiSNA, Navarra Institute for Health Research, Pamplona, Spain
| | - Chiara Ambrogio
- Department of Molecular Biotechnology and Health Sciences, Molecular Biotechnology Centre, University of Torino, Turin, Italy
| | - Fernando Lecanda
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain
- Centro de Investigación Biomédica en Red de Cáncer (CIBERONC), Madrid, Spain
- IdiSNA, Navarra Institute for Health Research, Pamplona, Spain
- Department of Pathology, Anatomy and Physiology, University of Navarra, Pamplona, Spain
| | - Silve Vicent
- Program in Solid Tumours, University of Navarra, Centre of Applied Medical Research (CIMA), 55 Pio XII Avenue, 31008, Pamplona, Spain.
- Centro de Investigación Biomédica en Red de Cáncer (CIBERONC), Madrid, Spain.
- IdiSNA, Navarra Institute for Health Research, Pamplona, Spain.
- Department of Pathology, Anatomy and Physiology, University of Navarra, Pamplona, Spain.
| |
Collapse
|
|
58
|
Rodenas MC, Peñas-Martínez J, Pardo-Sánchez I, Zaragoza-Huesca D, Ortega-Sabater C, Peña-García J, Espín S, Ricote G, Montenegro S, Ayala-De La Peña F, Luengo-Gil G, Nieto A, García-Molina F, Vicente V, Bernardi F, Lozano ML, Mulero V, Pérez-Sánchez H, Carmona-Bayonas A, Martínez-Martínez I. Venetoclax is a potent hepsin inhibitor that reduces the metastatic and prothrombotic phenotypes of hepsin-expressing colorectal cancer cells. Front Mol Biosci 2023; 10:1182925. [PMID: 37275957 PMCID: PMC10235687 DOI: 10.3389/fmolb.2023.1182925] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Grants] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 03/09/2023] [Accepted: 05/08/2023] [Indexed: 06/07/2023] Open
Abstract
Introduction: Hepsin is a type II transmembrane serine protease and its expression has been linked to greater tumorigenicity and worse prognosis in different tumors. Recently, our group demonstrated that high hepsin levels from primary tumor were associated with a higher risk of metastasis and thrombosis in localized colorectal cancer patients. This study aims to explore the molecular role of hepsin in colorectal cancer. Methods: Hepsin levels in plasma from resected and metastatic colorectal cancer patients were analyzed by ELISA. The effect of hepsin levels on cell migration, invasion, and proliferation, as well as on the activation of crucial cancer signaling pathways, was performed in vitro using colorectal cancer cells. A thrombin generation assay determined the procoagulant function of hepsin from these cells. A virtual screening of a database containing more than 2000 FDA-approved compounds was performed to screen hepsin inhibitors, and selected compounds were tested in vitro for their ability to suppress hepsin effects in colorectal cancer cells. Xenotransplantation assays were done in zebrafish larvae to study the impact of venetoclax on invasion promoted by hepsin. Results: Our results showed higher plasma hepsin levels in metastatic patients, among which, hepsin was higher in those suffering thrombosis. Hepsin overexpression increased colorectal cancer cell invasion, Erk1/2 and STAT3 phosphorylation, and thrombin generation in plasma. In addition, we identified venetoclax as a potent hepsin inhibitor that reduced the metastatic and prothrombotic phenotypes of hepsin-expressing colorectal cancer cells. Interestingly, pretreatment with Venetoclax of cells overexpressing hepsin reduced their invasiveness in vivo. Discussion: Our results demonstrate that hepsin overexpression correlates with a more aggressive and prothrombotic tumor phenotype. Likewise, they demonstrate the antitumor role of venetoclax as a hepsin inhibitor, laying the groundwork for molecular-targeted therapy for colorectal cancer.
Collapse
Affiliation(s)
- Maria Carmen Rodenas
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Julia Peñas-Martínez
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Irene Pardo-Sánchez
- Department of Cell Biology, Faculty of Biology, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - David Zaragoza-Huesca
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Carmen Ortega-Sabater
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Jorge Peña-García
- Computer Engineering Department, Structural Bioinformatics and High Performance Computing Research Group (BIO-HPC), UCAM Universidad Católica de Murcia, Guadalupe, Spain
| | - Salvador Espín
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Guillermo Ricote
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Sofía Montenegro
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Francisco Ayala-De La Peña
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Ginés Luengo-Gil
- Clinical Analysis and Pathology Department, Group of Molecular Pathology and Pharmacogenetics, IMIB-Pascual Parrilla, Hospital Universitario Santa Lucía, Cartagena, Spain
| | - Andrés Nieto
- Department of Pathology, Hospital Universitario Morales Meseguer, Murcia, Spain
| | | | - Vicente Vicente
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Francesco Bernardi
- Department of Life Sciences and Biotechnology, University of Ferrara, Ferrara, Italy
| | - María Luisa Lozano
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Victoriano Mulero
- Department of Cell Biology, Faculty of Biology, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Horacio Pérez-Sánchez
- Computer Engineering Department, Structural Bioinformatics and High Performance Computing Research Group (BIO-HPC), UCAM Universidad Católica de Murcia, Guadalupe, Spain
| | - Alberto Carmona-Bayonas
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| | - Irene Martínez-Martínez
- Department of Hematology and Medical Oncology, Hospital Universitario Morales Meseguer, Centro Regional de Hemodonación, Centro de Investigación Biomédica en Red de Enfermedades Raras, IMIB-Pascual Parrilla, Universidad de Murcia, Murcia, Spain
| |
Collapse
|
|
59
|
Zhu WT, Zeng XF, Yang H, Jia ML, Zhang W, Liu W, Liu SY. Resveratrol Loaded by Folate-Modified Liposomes Inhibits Osteosarcoma Growth and Lung Metastasis via Regulating JAK2/STAT3 Pathway. Int J Nanomedicine 2023; 18:2677-2691. [PMID: 37228445 PMCID: PMC10204760 DOI: 10.2147/ijn.s398046] [Citation(s) in RCA: 2] [Impact Index Per Article: 2.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/05/2023] [Accepted: 04/14/2023] [Indexed: 05/27/2023] Open
Abstract
Background Osteosarcoma is a malignant bone tumor with a high rate of lung metastasis and mortality. It has been demonstrated that resveratrol can inhibit tumor proliferation and metastasis, but its application is limited due to poor water solubility and low bioavailability. In this study, we proposed to prepare folate-modified liposomes loaded with resveratrol to investigate its anti-osteosarcoma effect in vitro and in vivo. Methods We prepared and characterized resveratrol liposomes modified with folate (denoted as, FA-Res/Lps). The effects of FA-Res/Lps on human osteosarcoma cell 143B proliferation, apoptosis, and migration were investigated by MTT, cell cloning, wound-healing assay, transwell, and flow cytometry. A xenograft tumor and lung metastasis model of osteosarcoma was constructed to study the therapeutic effects of FA-Res/Lps on the growth and metastasis of osteosarcoma in vivo. Results The FA-Res/Lps were prepared with a particle size of 118.5 ± 0.71 and a small dispersion coefficient of 0.154 ± 0.005. We found that FA-modified liposomes significantly increased resveratrol uptake by osteosarcoma cells 143B in flow cytometric assay, resulting in FA-Res/Lps, which inhibit tumor proliferation, migration and induce apoptosis more effectively than free Res and Res/Lps. The mechanism of action may be associated with the inhibition of JAK2/STAT3 signaling. In vivo imaging demonstrated that FA-modified DiR-modified liposomes significantly increased the distribution of drugs at the tumor site, leading to significant inhibition of osteosarcoma growth and metastasis by FA-Res/Lps. Furthermore, we found that FA-Res/Lps did not cause any adverse effects on mice body weight, liver, or kidney tissues. Conclusion Taken together, the anti-osteosarcoma effect of resveratrol is significantly enhanced when it is loaded into FA-modified liposomes. FA-Res/Lps is a promising strategy for the treatment of osteosarcoma.
Collapse
Affiliation(s)
- Wen Ting Zhu
- Department of Pharmacy, Biomedicine Research Center, Guangdong Provincial Key Laboratory of Major Obstetric Diseases, The Third Affiliated Hospital of Guangzhou Medical University, Guangzhou, 510150, People’s Republic of China
| | - Xiang Feng Zeng
- Department of Orthopedics, The Affiliated Nanhua Hospital, Hengyang Medical School, University of South China, Hengyang, 421001, People’s Republic of China
| | - Hua Yang
- Department of Orthopedics, The Affiliated Nanhua Hospital, Hengyang Medical School, University of South China, Hengyang, 421001, People’s Republic of China
| | - Meng Lei Jia
- Department of Pharmacy, Biomedicine Research Center, Guangdong Provincial Key Laboratory of Major Obstetric Diseases, The Third Affiliated Hospital of Guangzhou Medical University, Guangzhou, 510150, People’s Republic of China
| | - Wei Zhang
- Department of Orthopedics, The Affiliated Nanhua Hospital, Hengyang Medical School, University of South China, Hengyang, 421001, People’s Republic of China
| | - Wei Liu
- Department of Orthopedics, The Affiliated Nanhua Hospital, Hengyang Medical School, University of South China, Hengyang, 421001, People’s Republic of China
| | - Sheng Yao Liu
- Department of Orthopedics, The Second Affiliated Hospital of Guangzhou Medical University, Guangzhou, 510260, People’s Republic of China
| |
Collapse
|
|
60
|
Xuan SH, Hua ML, Xiang Z, He XL, Huang L, Jiang C, Dong P, Wu J. Roles of cancer stem cells in gastrointestinal cancers. World J Stem Cells 2023; 15:209-220. [PMID: 37181004 PMCID: PMC10173810 DOI: 10.4252/wjsc.v15.i4.209] [Citation(s) in RCA: 1] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Journal Information] [Submit a Manuscript] [Subscribe] [Scholar Register] [Received: 01/02/2023] [Revised: 02/25/2023] [Accepted: 03/27/2023] [Indexed: 04/26/2023] Open
Abstract
Cancer stem cells (CSCs) are the main cause of tumor growth, invasion, metastasis and recurrence. Recently, CSCs have been extensively studied to identify CSC-specific surface markers as well as signaling pathways that play key roles in CSCs self-renewal. The involvement of CSCs in the pathogenesis of gastrointestinal (GI) cancers also highlights these cells as a priority target for therapy. The diagnosis, prognosis and treatment of GI cancer have always been a focus of attention. Therefore, the potential application of CSCs in GI cancers is receiving increasing attention. This review summarizes the role of CSCs in GI cancers, focusing on esophageal cancer, gastric cancer, liver cancer, colorectal cancer, and pancreatic cancer. In addition, we propose CSCs as potential targets and therapeutic strategies for the effective treatment of GI cancers, which may provide better guidance for clinical treatment of GI cancers.
Collapse
Affiliation(s)
- Shi-Hai Xuan
- Department of Laboratory Medicine, The People’s Hospital of Dongtai City, Dongtai 224299, Jiangsu Province, China
| | - Meng-Lu Hua
- School of Medicine, Zhejiang University, Hangzhou 310009, Zhejiang Province, China
| | - Ze Xiang
- School of Medicine, Zhejiang University, Hangzhou 310009, Zhejiang Province, China
| | - Xiang-Lin He
- School of Medicine, Zhejiang University, Hangzhou 310009, Zhejiang Province, China
| | - Lan Huang
- Department of Clinical Laboratory, Suzhou Municipal Hospital, Suzhou 215008, Jiangsu Province, China
| | - Chun Jiang
- Department of Clinical Laboratory, Suzhou Municipal Hospital, Suzhou 215008, Jiangsu Province, China
| | - Peng Dong
- Hangzhou Institute of Cardiovascular Diseases, Hangzhou Normal University, Hangzhou 310015, Zhejiang Province, China
| | - Jian Wu
- Department of Clinical Laboratory, Suzhou Municipal Hospital, Suzhou 215008, Jiangsu Province, China
| |
Collapse
|
|
61
|
Duan L, Huang J, Zhang Y, Pi G, Ying X, Zeng F, Hu D, Ma J. FOXK1 regulates epithelial-mesenchymal transition and radiation sensitivity in nasopharyngeal carcinoma via the JAK/STAT3 signaling pathway. Genes Genomics 2023; 45:749-761. [PMID: 37043129 DOI: 10.1007/s13258-023-01380-y] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Grants] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/24/2022] [Accepted: 03/21/2023] [Indexed: 04/13/2023]
Abstract
BACKGROUND Nasopharyngeal carcinoma (NPC) is the most common head and neck tumor in China. Forkhead box (FOX) proteins have 19 subfamilies, which can maintain cell metabolism, regulate cell cycle and cell growth, etc. FOXK1 is a member of the FOX family, and studies have found that FOXK1 is closely related to tumors. OBJECTIVE This experiment aims to study the effects of FOXK1 interference on proliferation, apoptosis, invasion, epithelial-mesenchymal transition (EMT), and radiosensitivity, by regulating the Janus kinas/signal translator and activator of the transfer 3 (JAK/STAT3) pathway. METHODS The expression of FOXK1 was detected via immunohistochemistry using clinical nasopharyngeal carcinoma tissues and adjacent tissues. The relationship between FOXK1 expression and tumor stage was subsequently evaluated. The colony formation rate was calculated through the colony formation experiment. Cell apoptosis and cell cycle distribution were detected using flow cytometry, while cell invasion was detected using the Transwell method. The number of cells in the nucleus of each group after 30 min, 4 h, and 24 h of radiotherapy with the 2 Gy dose was counted using immunofluorescence under γ-H2AX focal points of a laser confocal microscope. RESULTS FOXK1 is clearly expressed in the patients' cancer tissues. The expression of FOXK1 was significantly correlated with the patient's sex. FOXK1 interference or Peficitinib can upregulate the apoptosis rate of 5-8 F and CNE-2 cells; increase the G2 phase of cells; and inhibit the invasion, migration, and EMT of cells. At the same time, FOXK1 interference can downregulate the protein expression of p-JAK1, p-JAK2, and p-STAT3 in cells. Interference from FOXK1 or Peficitinib alone can reduce the rate of cell colony formation under different radiation doses, and enhance the green fluorescence intensity of γ-H2AX in the nucleus after 4 and 24 h of the 2 Gy dose of radiotherapy. These results are optimal when FOXK1 interference and Peficitinib are used together. CONCLUSION FOXK1 interference in NPC cells can regulate EMT through the JAK/STAT3 signal pathway, enhance the radiosensitivity of cells, and thus inhibit tumor cell progression.
Collapse
Affiliation(s)
- Liqun Duan
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
| | - Jinlong Huang
- Department of Cardiology, People's Hospital of Dongxihu District, Wuhan, China
| | - Yong Zhang
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
| | - Guoliang Pi
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
| | - Xiaofang Ying
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
| | - Fanyu Zeng
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
| | - Desheng Hu
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China
| | - Jia Ma
- Department of Radiation Oncology, Hubei Cancer Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, China.
| |
Collapse
|
|
62
|
Pan L, Wu Q, Wang Y, Ma S, Zhang S. Characterization and mechanisms of radioresistant lung squamous cell carcinoma cell lines. Thorac Cancer 2023; 14:1239-1250. [PMID: 37028947 PMCID: PMC10175036 DOI: 10.1111/1759-7714.14865] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 12/17/2022] [Revised: 03/07/2023] [Accepted: 03/08/2023] [Indexed: 04/09/2023] Open
Abstract
BACKGROUND Radiotherapy is an important clinical treatment for patients with lung squamous cell carcinoma (LUSC), and resistance to radiotherapy is an important cause of recurrence and metastasis in LUSC. The aim of this study was to establish and explore the biological characteristics of radioresistant LUSC cells. MATERIALS AND METHODS The LUSC cell lines NCI-H2170 and NCI-H520 were irradiated (4 Gy × 15Fraction). Radiosensitivity, cell apoptosis, cell cycle, and DNA damage repair were measured by clonogenic survival assay, flow cytometry, immunofluorescence for γ-H2AX foci, and Comet assay, respectively. Activation of p-ATM(Ser1981), p-CHK2(Th68), p-DNA-PKcs (Ser2056), and Ku70/Ku80 was measured by western blot. Proteomics was used to explore the differential genes and enriched signaling pathways between radioresistant cell lines and parental lines. In vivo nude mouse xenograft experiments further verified the feasibility of the radioresistant LUSC cell lines. RESULTS After fractionated irradiation (total dose of 60 Gy), radioresistant cells had decreased radiosensitivity, increased G0/G1 phase arrest, enhanced DNA damage repair ability, and through the ATM/CHK2 and DNA-PKcs/Ku70 pathways regulated double strands break. The upregulated differential genes in radioresistant cell lines were mainly enriched in biological pathways such as cell migration and extracellular matrix (ECM)-receptor interaction. In vivo verification of decreased radiosensitivity of radioresistant cells CONCLUSIONS: Radioresistant LUSC cell lines were established by fractional radiotherapy, which regulates IR-induced DNA damage repair through ATM/CHK2 and DNA-PKcs/Ku70. Tandem Mass Tags (TMT) quantitative proteomics found that the biological process pathway of cell migration and ECM-receptor interaction are upregulated in LUSC radioresistant cells.
Collapse
Affiliation(s)
- Lifang Pan
- Department of the Fourth Clinical Medical College, Zhejiang Chinese Medical University, Hangzhou, China
| | - Qiong Wu
- Department of Integrated Chinese and Western Medicine, The Cancer Hospital of the University of Chinese Academy of Sciences (Zhejiang Cancer Hospital), Hangzhou, China
| | - Yuqing Wang
- Department of Translational Medicine Research Center, Key Laboratory of Clinical Cancer Pharmacology and Toxicology Research of Zhejiang Province, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine, Hangzhou, China
| | - Shenglin Ma
- Department of the Fourth Clinical Medical College, Zhejiang Chinese Medical University, Hangzhou, China
- Department of Oncology, Affiliated Hangzhou Cancer Hospital, Zhejiang University School of Medicine, Hangzhou, China
| | - Shirong Zhang
- Department of Translational Medicine Research Center, Key Laboratory of Clinical Cancer Pharmacology and Toxicology Research of Zhejiang Province, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine, Hangzhou, China
| |
Collapse
|
|
63
|
Mezher M, Abdallah S, Ashekyan O, Shoukari AA, Choubassy H, Kurdi A, Temraz S, Nasr R. Insights on the Biomarker Potential of Exosomal Non-Coding RNAs in Colorectal Cancer: An In Silico Characterization of Related Exosomal lncRNA/circRNA–miRNA–Target Axis. Cells 2023; 12:cells12071081. [PMID: 37048155 PMCID: PMC10093117 DOI: 10.3390/cells12071081] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/26/2023] [Revised: 03/28/2023] [Accepted: 03/31/2023] [Indexed: 04/07/2023] Open
Abstract
Colorectal cancer (CRC) is one of the most common cancer types, ranking third after lung and breast cancers. As such, it demands special attention for better characterization, which may eventually result in the development of early detection strategies and preventive measures. Currently, components of bodily fluids, which may reflect various disease states, are being increasingly researched for their biomarker potential. One of these components is the circulating extracellular vesicles, namely, exosomes, which are demonstrated to carry various cargo. Of importance, the non-coding RNA cargo of circulating exosomes, especially long non-coding RNAs (lncRNAs), circular RNAs (circRNAs), and micro RNAs (miRNAs), may potentially serve as significant diagnostic and prognostic/predictive biomarkers. In this review, we present existing evidence on the diagnostic and prognostic/predictive biomarker value of exosomal non-coding RNAs in CRC. In addition, taking advantage of the miRNA sponging functionality of lncRNAs and circRNAs, we demonstrate an experimentally validated CRC exosomal non-coding RNA-regulated target gene axis benefiting from published miRNA sponging studies in CRC. Hence, we present a set of target genes and pathways downstream of the lncRNA/circRNA–miRNA–target axis along with associated significant Gene Ontology (GO) terms and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathways, which may collectively serve to better characterize CRC and shed light on the significance of exosomal non-coding RNAs in CRC diagnosis and prognosis/prediction.
Collapse
Affiliation(s)
- Maria Mezher
- Department of Internal Medicine, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
| | - Samira Abdallah
- Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
| | - Ohanes Ashekyan
- Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
- Department of Biochemistry and Molecular Genetics, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
| | - Ayman Al Shoukari
- Department of Experimental Pathology, Immunology, and Microbiology, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
| | - Hayat Choubassy
- Faculty of Sciences, Lebanese University, Beirut P.O. Box 6573, Lebanon
| | - Abdallah Kurdi
- Department of Biochemistry and Molecular Genetics, American University of Beirut, Beirut 1107 2020, Lebanon
| | - Sally Temraz
- Department of Internal Medicine, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
| | - Rihab Nasr
- Department of Anatomy, Cell Biology and Physiological Sciences, Faculty of Medicine, American University of Beirut, Beirut 1107 2020, Lebanon
| |
Collapse
|
|
64
|
Montoyo-Pujol YG, García-Escolano M, Ponce JJ, Delgado-García S, Martín TA, Ballester H, Castellón-Molla E, Martínez-Peinado P, Pascual-García S, Sempere-Ortells JM, Peiró G. Variable Intrinsic Expression of Immunoregulatory Biomarkers in Breast Cancer Cell Lines, Mammospheres, and Co-Cultures. Int J Mol Sci 2023; 24:4478. [PMID: 36901916 PMCID: PMC10003642 DOI: 10.3390/ijms24054478] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 11/28/2022] [Revised: 02/13/2023] [Accepted: 02/22/2023] [Indexed: 03/02/2023] Open
Abstract
Advances in immunotherapy have increased interest in knowing the role of the immune system in breast cancer (BC) pathogenesis. Therefore, immune checkpoints (IC) and other pathways related to immune regulation, such as JAK2 and FoXO1, have emerged as potential targets for BC treatment. However, their intrinsic gene expression in vitro has not been extensively studied in this neoplasia. Thus, we evaluated the mRNA expression of tumor-cell-intrinsic CTLA-4, PDCD1 (PD1), CD274 (PD-L1), PDCD1LG2 (PD-L2), CD276 (B7-H3), JAK2, and FoXO1 in different BC cell lines, derived mammospheres, and co-cultures with peripheral blood mononuclear cells (PBMCs) by real-time quantitative polymerase chain reaction (qRT-PCR). Our results showed that intrinsic CTLA-4, CD274 (PD-L1), and PDCD1LG2 (PD-L2) were highly expressed in triple-negative cell lines, while CD276 was predominantly overexpressed in luminal cell lines. In contrast, JAK2 and FoXO1 were under-expressed. Moreover, high levels of CTLA-4, PDCD1 (PD1), CD274 (PD-L1), PDCD1LG2 (PD-L2), and JAK2 were found after mammosphere formation. Finally, the interaction between BC cell lines and peripheral blood mononuclear cells (PBMCs) stimulates the intrinsic expression of CTLA-4, PCDC1 (PD1), CD274 (PD-L1), and PDCD1LG2 (PD-L2). In conclusion, the intrinsic expression of immunoregulatory genes seems very dynamic, depending on BC phenotype, culture conditions, and tumor-immune cell interactions.
Collapse
Affiliation(s)
- Yoel Genaro Montoyo-Pujol
- Research Unit, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
- Medical Oncology Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Marta García-Escolano
- Research Unit, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - José J. Ponce
- Medical Oncology Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Silvia Delgado-García
- Gynecology and Obstetrics Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Tina Aurora Martín
- Gynecology and Obstetrics Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Hortensia Ballester
- Gynecology and Obstetrics Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Elena Castellón-Molla
- Pathology Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Pascual Martínez-Peinado
- Biotechnology Department, Immunology Division, University of Alicante, Ctra San Vicente s/n., 03080 San Vicente del Raspeig, Spain
| | - Sandra Pascual-García
- Biotechnology Department, Immunology Division, University of Alicante, Ctra San Vicente s/n., 03080 San Vicente del Raspeig, Spain
| | - José Miguel Sempere-Ortells
- Biotechnology Department, Immunology Division, University of Alicante, Ctra San Vicente s/n., 03080 San Vicente del Raspeig, Spain
- Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
| | - Gloria Peiró
- Research Unit, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
- Pathology Department, Dr. Balmis University General Hospital, Alicante Institute for Health and Biomedical Research (ISABIAL), Pintor Baeza 12, 03010 Alicante, Spain
- Biotechnology Department, Immunology Division, University of Alicante, Ctra San Vicente s/n., 03080 San Vicente del Raspeig, Spain
| |
Collapse
|
|
65
|
Li X, Liu J. FANCD2 inhibits ferroptosis by regulating the JAK2/STAT3 pathway in osteosarcoma. BMC Cancer 2023; 23:179. [PMID: 36814203 PMCID: PMC9945409 DOI: 10.1186/s12885-023-10626-7] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/09/2022] [Accepted: 02/10/2023] [Indexed: 02/24/2023] Open
Abstract
BACKGROUND This research aimed to investigate the roles of fanconi anemia complementation group D2 (FANCD2) on the regulation of ferroptosis in osteosarcoma progression. METHODS The function of FANCD2 on cell viability, invasion, migration, and tumor growth were explored. FANCD2 and pathway-related genes were determined by western blot. Ferroptosis-associated markers were determined, including lipid peroxidation, labile iron pool (LIP), ferrous iron (Fe2+), and ferroptosis-related genes. RESULTS FANCD2 expression was increased in osteosarcoma cells. FANCD2 knockdown reduced cell viability, invasion, and migration of osteosarcoma cells. FANCD2 knockdown regulated ferroptosis-related gene expression, and distinctly increased the levels of LIP, Fe2+, and lipid peroxidation, and these effects were reversed by a ferroptosis inhibitor Fer-1. In addition, JAK2 and STAT3 expression were reduced by silencing of FANCD2, and STAT3 activator (colivelin) distinctly reversed tumor suppressor effects of FANCD2 silencing on osteosarcoma development. CONCLUSION These findings suggested that FANCD2 silencing could suppress osteosarcoma cell viability, migration, invasion, and tumor growth, and induced ferroptosis by regulating the JAK2/STAT3 axis. These findings may provide novel therapeutic ideas for clinical treatment of osteosarcoma.
Collapse
Affiliation(s)
- Xujun Li
- grid.8547.e0000 0001 0125 2443Department of Orthopaedic, Minhang Hospital, Fudan University, No.170, Xinsong Road, Xinzhuang Town, Minhang District, Shanghai City, 201199 China
| | - Jiangyi Liu
- Department of Orthopaedic, Minhang Hospital, Fudan University, No.170, Xinsong Road, Xinzhuang Town, Minhang District, Shanghai City, 201199, China.
| |
Collapse
|
|
66
|
Zhang H, Yang H, Wei W, Zhao J, Vijayalakshmi A, Wang M. Ethacridine Regulation of JAK/STAT/ERK Signaling Pathway in Colon Cancer Cells SW620: In Vitro Approach. Pharmacogn Mag 2023. [DOI: 10.1177/09731296221136906] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 02/09/2023] Open
Abstract
Background Ethacridine have anticancer effects by inhibiting regulatory transcription factors and cell viability in various cancer cells. To investigate the effect of ethacridine on colorectal cancer cell lines, SW620 was studied via regulation of JAK/STAT/ERK signaling pathway. Materials and Methods Different doses of ethacridine (5–35 µM) expressed antiproliferative effects by decreasing the viability in a dose-dependent manner and the IC50 value was found to be 10 µM. Results Subsequent treatment with 10 µM of ethacridine showed that it induced mitochondrial dysfunction and reactive oxygen species generation. DAPI and PI staining assays revealed prominent apoptotic cells under the microscope when treated with 10 µM of ethacridine. In the mRNA expression study performing RT-PCR of apoptotic markers, cyclin-D1, Bax, Bcl-2, caspase 3, C-Myc, and surviving, enhanced levels of these markers were suppressed, which was inversely proportional to the levels of apoptotic enhancers namely Bax and caspase-3. It was also observed that increased NF-κB, IL-6, TNF-α, and COX-2 in colorectal cancer are suppressed by ethacridine. The expressions of JAK/STAT/ERK were also significantly suppressed after ethacridine treatment in SW620 cell lines. Conclusion In summary, it was corroborated that ethacridine promoted apoptosis in colon cancer cells by inhibiting quite a few cell signaling factors.
Collapse
Affiliation(s)
- Hui Zhang
- Department of Gastroenterology, Hanzhong Central Hospital, Hanzhong, China
| | - Hui Yang
- Department of Oncology Rehabilitation, Shaanxi Kangfu Hospital, Xi’an, China
| | - Wei Wei
- Department of Pathology, Xi’an No. 3 Hospital, Xi’an, China
| | - Jing Zhao
- Department of Oncology Rehabilitation, Shaanxi Kangfu Hospital, Xi’an, China
| | - Annamalai Vijayalakshmi
- Department of Biochemistry, Rabiammal Ahamed Maideen College for Women, Thiruvarur, Tamil Nadu, India
| | - Minhong Wang
- Department of Digestive Oncology, Pucheng County Hospital, Weinan, China
| |
Collapse
|
|
67
|
Gao Y, Li Y, Lin Z, Zeng Y, Huang Z, Han L, Zhong Y, Gong Y, Wu Q, Xie C. Ataxia telangiectasia mutated kinase inhibition promotes irradiation-induced PD-L1 expression in tumour-associated macrophages through IFN-I/JAK signalling pathway. Immunology 2023; 168:346-361. [PMID: 36326481 DOI: 10.1111/imm.13602] [Citation(s) in RCA: 1] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 03/06/2022] [Accepted: 10/07/2022] [Indexed: 11/06/2022] Open
Abstract
Tumour-associated macrophages (TAMs) are one of the primary sources of PD-L1 expression in the tumour microenvironment (TME). Ionizing radiation (IR) promotes PD-L1 expression in tumour cells. However, the effect of IR on macrophage PD-L1 expression and the underlying mechanisms remain unclear. ATM kinase, as the key kinase for initiating DNA damage repair (DDR) process, is associated with innate immune STING axis activation. Here, we explored the molecular mechanism implicated in macrophage PD-L1 expression regulated by IR as well as the role of ATM kinase in this process. IR-regulated PD-L1 expression in macrophages and associated signalling pathways were explored by in vitro studies using murine and human macrophage cell lines. A colorectal xenograft murine model was employed to demonstrate the impact of targeting ATM and PD-L1 expression in TAMs following IR on growth of tumour in vivo. IR up-regulated PD-L1 expression in macrophages, which was further augmented by ATM kinase inhibition. ATM inhibition increased IR-induced DNA damage, which activated STING/interferon regulatory factor 3 (IRF3) signalling pathway and up-regulated type I interferon (IFN-I) expression in macrophages. IFN-I bound to the IFN α receptor 1 on macrophages, activated the downstream JAK1 and STAT1/3 signalling and eventually led to PD-L1 up-expression. ATM inhibition augmented IR-induced PD-L1 expression in macrophages and CD8+ T cell infiltration, and promoted anti-tumour efficacy in vivo. These results suggested that ATM inhibition promoted IR-induced PD-L1 expression through the activation of innate immunity in TAMs, which provided a novel approach to enhance the anti-tumour efficacy of RT.
Collapse
Affiliation(s)
- Yuke Gao
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Yangyi Li
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Zaihuan Lin
- Department of Gastric and Colorectal Surgical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Yuxin Zeng
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Zhengrong Huang
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China.,Department of Biological Repositories, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Linzhi Han
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Yahua Zhong
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China.,Hubei Key Laboratory of Tumor Biological Behaviors, Zhongnan Hospital of Wuhan University, Wuhan, China.,Hubei Cancer Clinical Study Center, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Yan Gong
- Department of Biological Repositories, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Qiuji Wu
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China.,Hubei Key Laboratory of Tumor Biological Behaviors, Zhongnan Hospital of Wuhan University, Wuhan, China.,Hubei Cancer Clinical Study Center, Zhongnan Hospital of Wuhan University, Wuhan, China
| | - Conghua Xie
- Department of Radiation and Medical Oncology, Zhongnan Hospital of Wuhan University, Wuhan, China.,Hubei Key Laboratory of Tumor Biological Behaviors, Zhongnan Hospital of Wuhan University, Wuhan, China.,Hubei Cancer Clinical Study Center, Zhongnan Hospital of Wuhan University, Wuhan, China
| |
Collapse
|
|
68
|
Yuan H, Gui R, Wang Z, Fang F, Zhao H. Gut microbiota: A novel and potential target for radioimmunotherapy in colorectal cancer. Front Immunol 2023; 14:1128774. [PMID: 36798129 PMCID: PMC9927011 DOI: 10.3389/fimmu.2023.1128774] [Citation(s) in RCA: 1] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 12/21/2022] [Accepted: 01/17/2023] [Indexed: 02/04/2023] Open
Abstract
Colorectal cancer (CRC) is one of the most common cancers, with a high mortality rate, and is a major burden on human health worldwide. Gut microbiota regulate human immunity and metabolism through producing numerous metabolites, which act as signaling molecules and substrates for metabolic reactions in various biological processes. The importance of host-gut microbiota interactions in immunometabolic mechanisms in CRC is increasingly recognized, and interest in modulating the microbiota to improve patient's response to therapy has been raising. However, the specific mechanisms by which gut microbiota interact with immunotherapy and radiotherapy remain incongruent. Here we review recent advances and discuss the feasibility of gut microbiota as a regulatory target to enhance the immunogenicity of CRC, improve the radiosensitivity of colorectal tumor cells and ameliorate complications such as radiotoxicity. Currently, great breakthroughs in the treatment of non-small cell lung cancer and others have been achieved by radioimmunotherapy, but radioimmunotherapy alone has not been effective in CRC patients. By summarizing the recent preclinical and clinical evidence and considering regulatory roles played by microflora in the gut, such as anti-tumor immunity, we discuss the potential of targeting gut microbiota to enhance the efficacy of radioimmunotherapy in CRC and expect this review can provide references and fresh ideas for the clinical application of this novel strategy.
Collapse
Affiliation(s)
- Hanghang Yuan
- Department of Nuclear Medicine, The First Hospital of Jilin University, Changchun, China,National Health Commission (NHC) Key laboratory of Radiobiology, School of Public Health, Jilin University, Changchun, China
| | - Ruirui Gui
- Department of Nuclear Medicine, The First Hospital of Jilin University, Changchun, China,National Health Commission (NHC) Key laboratory of Radiobiology, School of Public Health, Jilin University, Changchun, China
| | - Zhicheng Wang
- National Health Commission (NHC) Key laboratory of Radiobiology, School of Public Health, Jilin University, Changchun, China
| | - Fang Fang
- National Health Commission (NHC) Key laboratory of Radiobiology, School of Public Health, Jilin University, Changchun, China,*Correspondence: Fang Fang, ; Hongguang Zhao,
| | - Hongguang Zhao
- Department of Nuclear Medicine, The First Hospital of Jilin University, Changchun, China,*Correspondence: Fang Fang, ; Hongguang Zhao,
| |
Collapse
|
|
69
|
Wang XY, Beeraka NM, Xue NN, Yu HM, Yang Y, Liu MX, Nikolenko VN, Liu JQ, Zhao D. Identification of a three-gene prognostic signature for radioresistant esophageal squamous cell carcinoma. World J Clin Oncol 2023; 14:13-26. [PMID: 36699628 PMCID: PMC9850665 DOI: 10.5306/wjco.v14.i1.13] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Journal Information] [Submit a Manuscript] [Subscribe] [Scholar Register] [Received: 07/25/2022] [Revised: 10/25/2022] [Accepted: 12/06/2022] [Indexed: 01/10/2023] Open
Abstract
BACKGROUND Esophageal squamous cell carcinoma (ESCC) is causing a high mortality rate due to the lack of efficient early prognosis markers and suitable therapeutic regimens. The prognostic role of genes responsible for the acquisition of radioresistance in ESCC has not been fully elucidated.
AIM To establish a prognostic model by studying gene expression patterns pertinent to radioresistance in ESCC patients.
METHODS Datasets were obtained from the Gene Expression Omnibus and The Cancer Genome Atlas databases. The edgeR, a Bioconductor package, was used to analyze mRNA expression between different groups. We screened genes specifically responsible for radioresistance to estimate overall survival. Pearson correlation analysis was performed to confirm whether the expression of those genes correlated with each other. Genes contributing to radioresistance and overall survival were assessed by the multivariate Cox regression model through the calculation of βi and risk score using the following formula: 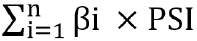 . .
RESULTS We identified three prognostic mRNAs (cathepsin S [CTSS], cluster of differentiation 180 [CD180], and SLP adapter and CSK-interacting membrane protein [SCIMP]) indicative of radioresistance. The expression of the three identified mRNAs was related to each other (r > 0.70 and P < 0.05). As to 1-year and 3-year overall survival prediction, the area under the time-dependent receiver operating characteristic curve of the signature consisting of the three mRNAs was 0.716 and 0.841, respectively. When stratifying patients based on the risk score derived from the signature, the high-risk group exhibited a higher death risk and shorter survival time than the low-risk group (P < 0.0001). Overall survival of the low-risk patients was significantly better than that of the high-risk patients (P = 0.018).
CONCLUSION We have developed a novel three-gene prognostic signature consisting of CTSS, CD180, and SCIMO for ESCC, which may facilitate the prediction of early prognosis of this malignancy.
Collapse
Affiliation(s)
- Xiao-Yan Wang
- Department of Endocrinology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Narasimha M Beeraka
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
- Department of Human Anatomy, I. M. Sechenov First Moscow State Medical University, Moscow 119991, Russia
- Department of Pharmaceutical Chemistry, JSS College of Pharmacy, Mysuru 570015, India
| | - Nan-Nan Xue
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Hui-Ming Yu
- Department of Radiation Oncology, Peking University Cancer Hospital & Institute, Beijing 065005, China
| | - Ya Yang
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Mao-Xing Liu
- Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education), Department of Gastrointestinal Surgery IV, Peking University Cancer Hospital & Institute, Beijing, China
| | - Vladimir N Nikolenko
- Department of Human Anatomy, I. M. Sechenov First Moscow State Medical University, Moscow 119991, Russia
- M.V. Lomonosov Moscow State University, Moscow 119991, Russia
| | - Jun-Qi Liu
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Di Zhao
- Department of Endocrinology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| |
Collapse
|
|
70
|
Zhang C, Zhao S, Deng H, Zhang S, Wang J, Song X, Yu D, Zhang Y, Deng W. STAT3 promotes RNA polymerase III-directed transcription by controlling the miR-106a-5p/TP73 axis. eLife 2023; 12:e82826. [PMID: 36656267 PMCID: PMC9851613 DOI: 10.7554/elife.82826] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 08/18/2022] [Accepted: 01/05/2023] [Indexed: 01/20/2023] Open
Abstract
Deregulation of Pol III products causes a range of diseases, including neural diseases and cancers. However, the factors and mechanisms that modulate Pol III-directed transcription remain to be found, although massive advances have been achieved. Here, we show that STAT3 positively regulates the activities of Pol III-dependent transcription and cancer cell growth. RNA-seq analysis revealed that STAT3 inhibits the expression of TP73, a member of the p53 family. We found that TP73 is not only required for the regulation of Pol III-directed transcription mediated by STAT3 but also independently suppresses the synthesis of Pol III products. Mechanistically, TP73 can disrupt the assembly of TFIIIB subunits and inhibit their occupancies at Pol III target loci by interacting with TFIIIB subunit TBP. MiR-106a-5p can activate Pol III-directed transcription by targeting the TP73 mRNA 3' UTR to reduce TP 73 expression. We show that STAT3 activates the expression of miR-106a-5p by binding to the miRNA promoter, indicating that the miR-106a-5p links STAT3 with TP73 to regulate Pol III-directed transcription. Collectively, these findings indicate that STAT3 functions as a positive regulator in Pol III-directed transcription by controlling the miR-106a-5p/TP73 axis.
Collapse
Affiliation(s)
- Cheng Zhang
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Shasha Zhao
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Huan Deng
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Shihua Zhang
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Juan Wang
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
- School of Materials and Metallurgy, Wuhan University of Science and TechnologyWuhanChina
| | - Xiaoye Song
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Deen Yu
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Yue Zhang
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| | - Wensheng Deng
- School of Life Science and Health, Wuhan University of Science and TechnologyWuhanChina
| |
Collapse
|
|
71
|
Li W, Wei W, Hu D, Tang R, Hu Z. circRNA ITGA7 restrains growth and enhances radiosensitivity by up-regulating SMAD4 in colorectal carcinoma. Open Med (Wars) 2023; 18:20220604. [PMID: 36694626 PMCID: PMC9835197 DOI: 10.1515/med-2022-0604] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/18/2022] [Revised: 09/23/2022] [Accepted: 10/18/2022] [Indexed: 01/13/2023] Open
Abstract
Circular RNAs have been reported to be widely involved in cancer cell tumorigenesis and drug resistance; here, the aim of this study was to investigate whether circRNA Integrin Subunit Alpha 7 (ITGA7) (circ_ITGA7) was associated with the tumor growth and radiosensitivity of colorectal cancer (CRC). We found that circ_ITGA7 expression was lower in CRC tissues and cells than those in the normal tissues and cell lines according to quantitative real-time polymerase chain reaction. As shown by cell counting kit-8 assay, flow cytometry, colony formation assay, and xenograft experiment, ectopic overexpression of circ_ITGA7 remarkably restrained CRC tumor growth and enhanced radiosensitivity in vitro and in vivo. Mechanistically, circ_ITGA7 could target microRNA (miR)-766 to prevent the degradation of its target gene mothers against decapentaplegic homolog 4 (SMAD4), the binding between miR-766 and circ_ITGA7 or SMAD4 was first verified by dual-luciferase activity assay. Additionally, miR-766 up-regulation reversed the inhibitory effects of circ_ITGA7 on CRC growth and radiosensitivity. Besides that, inhibition of miR-766 reduced CRC cell growth and sensitized cells to radiotherapy, and these effects mediated by miR-766 inhibitor were rescued by the silencing of SMAD4. In all, circ_ITGA7 suppressed CRC growth and enhanced radiosensitivity by up-regulating SMAD4 through sequestering miR-766, providing an insight for the further development of CRC treatment.
Collapse
Affiliation(s)
- Wei Li
- Department of Anorectal Hemorrhoids, Gaozhou People’s Hospital, Gaozhou City, Guangdong Province, 525200China
| | - Wancheng Wei
- Department of Anorectal Hemorrhoids, Gaozhou People’s Hospital, Gaozhou City, Guangdong Province, 525200China
| | - Dingyin Hu
- Department of Anorectal Hemorrhoids, Gaozhou People’s Hospital, Gaozhou City, Guangdong Province, 525200China
| | - Rutong Tang
- Department of Anorectal Hemorrhoids, Gaozhou People’s Hospital, Gaozhou City, Guangdong Province, 525200China
| | - Zikang Hu
- Department of Anorectal Hemorrhoids, Gaozhou People’s Hospital, No. 89, Xiguan Road, Gaozhou City, Guangdong Province, 525200China
| |
Collapse
|
|
72
|
Tessmann JW, Rocha MR, Morgado-Díaz JA. Mechanisms of radioresistance and the underlying signaling pathways in colorectal cancer cells. J Cell Biochem 2023; 124:31-45. [PMID: 36565460 DOI: 10.1002/jcb.30361] [Citation(s) in RCA: 2] [Impact Index Per Article: 2.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/27/2022] [Revised: 11/23/2022] [Accepted: 12/13/2022] [Indexed: 12/25/2022]
Abstract
Radiotherapy is one of the most common modalities for the treatment of a wide range of tumors, including colorectal cancer (CRC); however, radioresistance of cancer cells remains a major limitation for this treatment. Following radiotherapy, the activities of various cellular mechanisms and cell signaling pathways are altered, resulting in the development of radioresistance, which leads to therapeutic failure and poor prognosis in patients with cancer. Furthermore, even though several inhibitors have been developed to target tumor resistance, these molecules can induce side effects in nontumor cells due to low specificity and efficiency. However, the role of these mechanisms in CRC has not been extensively studied. This review discusses recent studies regarding the relationship between radioresistance and the alterations in a series of cellular mechanisms and cell signaling pathways that lead to therapeutic failure and tumor recurrence. Our review also presents recent advances in the in vitro/in vivo study models aimed at investigating the radioresistance mechanism in CRC. Furthermore, it provides a relevant biochemical basis in theory, which can be useful to improve radiotherapy sensitivity and prolong patient survival.
Collapse
Affiliation(s)
- Josiane W Tessmann
- Cellular and Molecular Oncobiology Program, Brazilian National Cancer Institute (INCA), Rio de Janeiro, Brazil
| | - Murilo R Rocha
- Cellular and Molecular Oncobiology Program, Brazilian National Cancer Institute (INCA), Rio de Janeiro, Brazil
| | - Jose A Morgado-Díaz
- Cellular and Molecular Oncobiology Program, Brazilian National Cancer Institute (INCA), Rio de Janeiro, Brazil
| |
Collapse
|
|
73
|
Zhu M, Li S, Cao X, Rashid K, Liu T. The STAT family: Key transcription factors mediating crosstalk between cancer stem cells and tumor immune microenvironment. Semin Cancer Biol 2023; 88:18-31. [PMID: 36410636 DOI: 10.1016/j.semcancer.2022.11.011] [Citation(s) in RCA: 8] [Impact Index Per Article: 8.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 06/03/2022] [Revised: 11/14/2022] [Accepted: 11/16/2022] [Indexed: 11/21/2022]
Abstract
Signal transducer and activator of transcription (STAT) proteins compose a family of transcription factors critical for cancer stem cells (CSCs), and they are involved in maintaining stemness properties, enhancing cell proliferation, and promoting metastasis. Recent studies suggest that STAT proteins engage in reciprocal communication between CSCs and infiltrate immune cell populations in the tumor microenvironment (TME). Emerging evidence has substantiated the influence of immune cells, including macrophages, myeloid-derived suppressor cells, and T cells, on CSC survival through the regulation of STAT signaling. Conversely, dysregulation of STATs in CSCs or immune cells contributes to the establishment of an immunosuppressive TME. Thus, STAT proteins are promising therapeutic targets for cancer treatment, especially when used in combination with immunotherapy. From this perspective, we discuss the complex roles of STATs in CSCs and highlight their functions in the crosstalk between CSCs and the immune microenvironment. Finally, cutting-edge clinical trial progress with STAT signaling inhibitors is summarized.
Collapse
Affiliation(s)
- Mengxuan Zhu
- Department of Medical Oncology, Zhongshan Hospital, Fudan University, Shanghai, China; Center of Evidence-based Medicine, Fudan University, Shanghai, China
| | - Suyao Li
- Department of Medical Oncology, Zhongshan Hospital, Fudan University, Shanghai, China; Center of Evidence-based Medicine, Fudan University, Shanghai, China
| | - Xin Cao
- Institute of Clinical Science, Zhongshan Hospital, Fudan University, Shanghai, China.
| | - Khalid Rashid
- Department of Cancer Biology, Faculty of Medicine, University of Cincinnati, OH, USA.
| | - Tianshu Liu
- Department of Medical Oncology, Zhongshan Hospital, Fudan University, Shanghai, China; Center of Evidence-based Medicine, Fudan University, Shanghai, China.
| |
Collapse
|
|
74
|
Huang A, Shi J, Sun Z, Yang Y, Gao Z, Gu J. Identification of a prognostic signature and ENTR1 as a prognostic biomarker for colorectal mucinous adenocarcinoma. Front Oncol 2023; 13:1061785. [PMID: 37182178 PMCID: PMC10172661 DOI: 10.3389/fonc.2023.1061785] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Grants] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/05/2022] [Accepted: 04/11/2023] [Indexed: 05/16/2023] Open
Abstract
Background Mucinous adenocarcinoma (MAC) is a unique clinicopathological colorectal cancer (CRC) type that has been recognized as a separate entity from non-mucinous adenocarcinoma (NMAC), with distinct clinical, pathologic, and molecular characteristics. We aimed to construct prognostic signatures and identifying candidate biomarkers for patients with MAC. Methods Differential expression analysis, weighted correlation network analysis (WGCNA), and least absolute shrinkage and selection operator (LASSO)-Cox regression model were used to identify hub genes and construct a prognostic signature based on RNA sequencing data from TCGA datasets. The Kaplan-Meier survival curve, gene set enrichment analysis (GSEA), cell stemness, and immune infiltration were analyzed. Biomarker expression in MAC and corresponding normal tissues from patients operated in 2020 was validated using immunohistochemistry. Results We constructed a prognostic signature based on ten hub genes. Patients in the high-risk group had significantly worse overall survival (OS) than patients in the low-risk group (p < 0.0001). We also found that ENTR1 was closely associated with OS (p = 0.016). ENTR1 expression was significantly positively correlated with cell stemness of MAC (p < 0.0001) and CD8+ T cell infiltration (p = 0.01), whereas it was negatively associated with stromal scores (p = 0.03). Finally, the higher expression of ENTR1 in MAC tissues than in normal tissues was validated. Conclusion We established the first MAC prognostic signature, and determined that ENTR1 could serve as a prognostic marker for MAC.
Collapse
Affiliation(s)
- An Huang
- Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Gastrointestinal Surgery III, Peking University Cancer Hospital & Institute, Beijing, China
| | - Jingyi Shi
- Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Gastrointestinal Surgery III, Peking University Cancer Hospital & Institute, Beijing, China
| | - Zhuang Sun
- Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Gastrointestinal Surgery III, Peking University Cancer Hospital & Institute, Beijing, China
| | - Yong Yang
- Department of Gastrointestinal Surgery, Peking University Shougang Hospital, Beijing, China
| | - Zhaoya Gao
- Department of Gastrointestinal Surgery, Peking University Shougang Hospital, Beijing, China
- Department of General Surgery, Peking University First Hospital, Beijing, China
| | - Jin Gu
- Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education/Beijing), Department of Gastrointestinal Surgery III, Peking University Cancer Hospital & Institute, Beijing, China
- Department of Gastrointestinal Surgery, Peking University Shougang Hospital, Beijing, China
- Peking Tsinghua Center for Life Science, Peking University International Cancer Center, Beijing, China
- *Correspondence: Jin Gu,
| |
Collapse
|
|
75
|
Xiong Y, Jian J, Yu H, Wu J, Mao H, Feng R, Wang L, Jian Y, Liu X. Cyanidin-3-O-glucoside plays a protective role against renal ischemia/ reperfusion injury via the JAK/STAT pathway. Acta Cir Bras 2023; 38:e381023. [PMID: 37132754 PMCID: PMC10158851 DOI: 10.1590/acb381023] [Citation(s) in RCA: 2] [Impact Index Per Article: 2.0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/31/2022] [Accepted: 01/05/2023] [Indexed: 05/04/2023] Open
Abstract
PURPOSE To investigate the role of cyanidin-3-O-glucoside (C3G) in renal ischemia/reperfusion (I/R) injury and the potential mechanisms. METHODS Mouse models were established by clamping the left renal vessels, and in vitro cellular models were established by hypoxic reoxygenation. RESULTS Renal dysfunction and tissue structural damage were significantly higher in the I/R group. After treatment with different concentrations of C3G, the levels of renal dysfunction and tissue structural damage decreased at different levels. And its protective effect was most pronounced at 200 mg/kg. The use of C3G reduced apoptosis as well as the expression of endoplasmic reticulum stress (ERS)-related proteins. Hypoxia/reoxygenation (H/R)-induced apoptosis and ERS are dependent on oxidative stress in vitro. In addition, both AG490 and C3G inhibited the activation of JAK/STAT pathway and attenuated oxidative stress, ischemia-induced apoptosis and ERS. CONCLUSIONS The results demonstrated that C3G blocked renal apoptosis and ERS protein expression by preventing reactive oxygen species (ROS) production after I/R via the JAK/STAT pathway, suggesting that C3G may be a potential therapeutic agent for renal I/R injury.
Collapse
Affiliation(s)
- Yufeng Xiong
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| | - Jun Jian
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| | - Honglin Yu
- University of Science and Technology of China, The First Affiliated Hospital - Department of Radiology - Hefei (Anhui), China
| | - Jiejun Wu
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| | - Hu Mao
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| | - Ruikang Feng
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| | - Lei Wang
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| | - Yonghong Jian
- Renmin Hospital of Wuhan University - Department of Nephrology - Wuhan (Hubei), China
| | - Xiuheng Liu
- Renmin Hospital of Wuhan University - Department of Urology - Wuhan (Hubei), China
- Renmin Hospital of Wuhan University - Institute of Urologic Disease - Wuhan (Hubei), China
| |
Collapse
|
|
76
|
Liao W, Zhang L, Chen X, Xiang J, Zheng Q, Chen N, Zhao M, Zhang G, Xiao X, Zhou G, Zeng J, Tang J. Targeting cancer stem cells and signalling pathways through phytochemicals: A promising approach against colorectal cancer. PHYTOMEDICINE : INTERNATIONAL JOURNAL OF PHYTOTHERAPY AND PHYTOPHARMACOLOGY 2023; 108:154524. [PMID: 36375238 DOI: 10.1016/j.phymed.2022.154524] [Citation(s) in RCA: 4] [Impact Index Per Article: 4.0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Subscribe] [Scholar Register] [Received: 06/15/2022] [Revised: 10/10/2022] [Accepted: 10/24/2022] [Indexed: 06/16/2023]
Abstract
BACKGROUND Cancer stem cells (CSCs) are strongly associated with high tumourigenicity, chemotherapy or radiotherapy resistance, and metastasis and recurrence, particularly in colorectal cancer (CRC). Therefore, targeting CSCs may be a promising approach. Recently, discovery and research on phytochemicals that effectively target colorectal CSCs have been gaining popularity because of their broad safety profile and multi-target and multi-pathway modes of action. PURPOSE This review aimed to elucidate and summarise the effects and mechanisms of phytochemicals with potential anti-CSC agents that could contribute to the better management of CRC. METHODS We reviewed PubMed, EMBASE, Web of Science, Ovid, ScienceDirect and China National Knowledge Infrastructure databases from the original publication date to March 2022 to review the mechanisms by which phytochemicals inhibit CRC progression by targeting CSCs and their key signalling pathways. Phytochemicals were classified and summarised based on the mechanisms of action. RESULTS We observed that phytochemicals could affect the biological properties of colorectal CSCs. Phytochemicals significantly inhibit self-renewal, migration, invasion, colony formation, and chemoresistance and induce apoptosis and differentiation of CSCs by regulating the Wnt/β-catenin pathway (e.g., diallyl trisulfide and genistein), the phosphatidylinositol-3-kinase/protein kinase B/mammalian target of rapamycin pathway (e.g., caffeic acid and piperlongumine), the neurogenic locus notch homolog protein pathway (e.g., honokiol, quercetin, and α-mangostin), the Janus kinase-signal transducer and activator of transcription pathway (e.g., curcumin, morin, and ursolic acid), and other key signalling pathways. It is worth noting that several phytochemicals, such as resveratrol, silibinin, evodiamine, and thymoquinone, highlight multi-target and multi-pathway effects in restraining the malignant biological behaviour of CSCs. CONCLUSIONS This review demonstrates the potential of targeted therapies for colorectal CSCs using phytochemicals. Phytochemicals could serve as novel therapeutic agents for CRC and aid in drug development.
Collapse
Affiliation(s)
- Wenhao Liao
- Department of Oncology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Lanlan Zhang
- State Key Laboratory Basis of Xinjiang Indigenous Medicinal Plants Resource Utilization, Key Laboratory of Plant Resources and Chemistry in Arid Regions, Xinjiang Technical Institute of Physics and Chemistry, Chinese Academy of Sciences, Urumqi 830011, China; University of Chinese Academy of Sciences, Beijing 100049, China
| | - Xian Chen
- Department of Pathology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Juyi Xiang
- Center for drug evaluation, National Medical Products Administration, Beijing 100022, China
| | - Qiao Zheng
- Department of Oncology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Nianzhi Chen
- Department of Oncology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Maoyuan Zhao
- Department of Oncology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Gang Zhang
- Department of Oncology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Xiaolin Xiao
- Department of Oncology, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China
| | - Gang Zhou
- Center for drug evaluation, National Medical Products Administration, Beijing 100022, China.
| | - Jinhao Zeng
- TCM Regulating Metabolic Diseases Key Laboratory of Sichuan Province, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China; Department of Geriatrics, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China.
| | - Jianyuan Tang
- TCM Regulating Metabolic Diseases Key Laboratory of Sichuan Province, Hospital of Chengdu University of Traditional Chinese Medicine, Chengdu 610072, China.
| |
Collapse
|
|
77
|
Huang B, Lang X, Li X. The role of IL-6/JAK2/STAT3 signaling pathway in cancers. Front Oncol 2022; 12:1023177. [PMID: 36591515 PMCID: PMC9800921 DOI: 10.3389/fonc.2022.1023177] [Citation(s) in RCA: 68] [Impact Index Per Article: 34.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 08/19/2022] [Accepted: 12/06/2022] [Indexed: 12/23/2022] Open
Abstract
Interleukin-6 (IL-6) is a pleiotropic cytokine involved in immune regulation. It can activate janus kinase 2 (JAK2)-signal transducer and activator of transcription 3 (STAT3) signaling pathway. As one of the important signal transduction pathways in cells, JAK2/STAT3 signaling pathway plays a critical role in cell proliferation and differentiation by affecting the activation state of downstream effector molecules. The activation of JAK2/STAT3 signaling pathway is involved in tumorigenesis and development. It contributes to the formation of tumor inflammatory microenvironment and is closely related to the occurrence and development of many human tumors. This article focuses on the relationship between IL-6/JAK2/STAT3 signaling pathway and liver cancer, breast cancer, colorectal cancer, gastric cancer, lung cancer, pancreatic cancer and ovarian cancer, hoping to provide references for the research of cancer treatment targeting key molecules in IL-6/JAK2/STAT3 signaling pathway.
Collapse
Affiliation(s)
- Bei Huang
- Operational Management Office, West China Second University Hospital, Sichuan University, Chengdu, China,Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, Chengdu, China
| | - Xiaoling Lang
- Operational Management Office, West China Second University Hospital, Sichuan University, Chengdu, China,Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, Chengdu, China,*Correspondence: Xiaoling Lang, ; Xihong Li,
| | - Xihong Li
- Key Laboratory of Birth Defects and Related Diseases of Women and Children (Sichuan University), Ministry of Education, Chengdu, China,Emergency Department, West China Second University Hospital, Sichuan University, Chengdu, China,*Correspondence: Xiaoling Lang, ; Xihong Li,
| |
Collapse
|
|
78
|
Nanoparticle-Mediated Delivery of STAT3 Inhibitors in the Treatment of Lung Cancer. Pharmaceutics 2022; 14:pharmaceutics14122787. [PMID: 36559280 PMCID: PMC9781630 DOI: 10.3390/pharmaceutics14122787] [Citation(s) in RCA: 4] [Impact Index Per Article: 2.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/11/2022] [Revised: 12/05/2022] [Accepted: 12/08/2022] [Indexed: 12/15/2022] Open
Abstract
Lung cancer is a common malignancy worldwide, with high morbidity and mortality. Signal transducer and activator of transcription 3 (STAT3) is an important transcription factor that not only regulates different hallmarks of cancer, such as tumorigenesis, cell proliferation, and metastasis but also regulates the occurrence and maintenance of cancer stem cells (CSCs). Abnormal STAT3 activity has been found in a variety of cancers, including lung cancer, and its phosphorylation level is associated with a poor prognosis of lung cancer. Therefore, the STAT3 pathway may represent a promising therapeutic target for the treatment of lung cancer. To date, various types of STAT3 inhibitors, including natural compounds, small molecules, and gene-based therapies, have been developed through direct and indirect strategies, although most of them are still in the preclinical or early clinical stages. One of the main obstacles to the development of STAT3 inhibitors is the lack of an effective targeted delivery system to improve their bioavailability and tumor targetability, failing to fully demonstrate their anti-tumor effects. In this review, we will summarize the recent advances in STAT3 targeting strategies, as well as the applications of nanoparticle-mediated targeted delivery of STAT3 inhibitors in the treatment of lung cancer.
Collapse
|
|
79
|
Jiang XF, Zhang BM, Du FQ, Guo JN, Wang D, Li YE, Deng SH, Cui BB, Liu YL. Exploring biomarkers for prognosis and neoadjuvant chemosensitivity in rectal cancer: Multi-omics and ctDNA sequencing collaboration. Front Immunol 2022; 13:1013828. [PMID: 36569844 PMCID: PMC9780298 DOI: 10.3389/fimmu.2022.1013828] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 08/10/2022] [Accepted: 11/23/2022] [Indexed: 12/13/2022] Open
Abstract
Introduction This study aimed to identified the key genes and sequencing metrics for predicting prognosis and efficacy of neoadjuvant chemotherapy (nCT) in rectal cancer (RC) based on genomic DNA sequencing in samples with different origin and multi-omics association database. Methods We collected 16 RC patients and obtained DNA sequencing data from cancer tissues and plasma cell-free DNA before and after nCT. Various gene variations were analyzed, including single nucleotide variants (SNV), copy number variation (CNV), tumor mutation burden (TMB), copy number instability (CNI) and mutant-allele tumor heterogeneity (MATH). We also identified genes by which CNV level can differentiate the response to nCT. The Cancer Genome Atlas database and the Clinical Proteomic Tumor Analysis Consortium database were used to further evaluate the specific role of therapeutic relevant genes and screen out the key genes in multi-omics levels. After the intersection of the screened genes from differential expression analysis, survival analysis and principal components analysis dimensionality reduction cluster analysis, the key genes were finally identified. Results The genes CNV level of principal component genes in baseline blood and cancer tissues could significantly distinguish the two groups of patients. The CNV of HSP90AA1, EGFR, SRC, MTOR, etc. were relatively gained in the better group compared with the poor group in baseline blood. The CNI and TMB was significantly different between the two groups. The increased expression of HSP90AA1, EGFR, and SRC was associated with increased sensitivity to multiple chemotherapeutic drugs. The nCT predictive score obtained by therapeutic relevant genes could be a potential prognostic indicator, and the combination with TMB could further refine prognostic prediction for patients. After a series of analysis in multi-omics association database, EGFR and HSP90AA1 with significant differences in multiple aspects were identified as the key predictive genes related to prognosis and the sensitivity of nCT. Discussion This work revealed that effective combined application and analysis in multi-omics data are critical to search for predictive biomarkers. The key genes EGFR and HSP90AA1 could serve as an effective biomarker to predict prognose and neoadjuvant chemosensitivity.
Collapse
Affiliation(s)
- Xiu-Feng Jiang
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China
| | - Bo-Miao Zhang
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China
| | - Fen-Qi Du
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China
| | - Jun-Nan Guo
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China
| | - Dan Wang
- Department of Neurology, The Second Affiliated Hospital of Qiqihar Medical University, Qiqihar, China
| | - Yi-En Li
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China
| | - Shen-Hui Deng
- Department of Anesthesiology, The Fourth Affiliated Hospital of Harbin Medical University, Harbin, China
| | - Bin-Bin Cui
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China,*Correspondence: Bin-Bin Cui, ; Yan-Long Liu,
| | - Yan-Long Liu
- Department of Colorectal Surgery, Harbin Medical University Cancer Hospital, Harbin, China,*Correspondence: Bin-Bin Cui, ; Yan-Long Liu,
| |
Collapse
|
|
80
|
Guerrero-Rodríguez SL, Mata-Cruz C, Pérez-Tapia SM, Velasco-Velázquez MA. Role of CD36 in cancer progression, stemness, and targeting. Front Cell Dev Biol 2022; 10:1079076. [PMID: 36568966 PMCID: PMC9772993 DOI: 10.3389/fcell.2022.1079076] [Citation(s) in RCA: 16] [Impact Index Per Article: 8.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/25/2022] [Accepted: 11/24/2022] [Indexed: 12/14/2022] Open
Abstract
CD36 is highly expressed in diverse tumor types and its expression correlates with advanced stages, poor prognosis, and reduced survival. In cancer cells, CD36: 1) increases fatty acid uptake, reprogramming lipid metabolism; 2) favors cancer cell proliferation, and 3) promotes epithelial-mesenchymal transition. Furthermore, CD36 expression correlates with the expression of cancer stem cell markers and CD36+ cancer cells display increased stemness functional properties, including clonogenicity, chemo- and radioresistance, and metastasis-initiating capability, suggesting CD36 is a marker of the cancer stem cell population. Thus, CD36 has been pointed as a potential therapeutic target in cancer. At present, at least three different types of molecules have been developed for reducing CD36-mediated functions: blocking monoclonal antibodies, small-molecule inhibitors, and compounds that knock-down CD36 expression. Herein, we review the role of CD36 in cancer progression, its participation in stemness control, as well as the efficacy of reported CD36 inhibitors in cancer cell cultures and animal models. Overall, the evidence compiled points that CD36 is a valid target for the development of new anti-cancer therapies.
Collapse
Affiliation(s)
| | - Cecilia Mata-Cruz
- Pharmacology Department, School of Medicine, Universidad Nacional Autónoma de México, Mexico City, Mexico,Graduate Program in Biochemical Sciences, Universidad Nacional Autónoma de México, Mexico City, Mexico
| | - Sonia M. Pérez-Tapia
- Research and Development in Biotherapeutics Unit, National School of Biological Sciences, National Polytechnic Institute, Mexico City, Mexico,National Laboratory for Specialized Services of Investigation Development and Innovation (I+D+i) for Pharma Chemicals and Biotechnological products LANSEIDI-FarBiotec-CONACyT, Mexico City, Mexico,Immunology Department, National School of Biological Sciences, National Polytechnic Institute, Mexico City, Mexico
| | - Marco A. Velasco-Velázquez
- Pharmacology Department, School of Medicine, Universidad Nacional Autónoma de México, Mexico City, Mexico,*Correspondence: Marco A. Velasco-Velázquez,
| |
Collapse
|
|
81
|
Ervin EH, French R, Chang CH, Pauklin S. Inside the stemness engine: Mechanistic links between deregulated transcription factors and stemness in cancer. Semin Cancer Biol 2022; 87:48-83. [PMID: 36347438 DOI: 10.1016/j.semcancer.2022.11.001] [Citation(s) in RCA: 1] [Impact Index Per Article: 0.5] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/13/2022] [Revised: 10/22/2022] [Accepted: 11/03/2022] [Indexed: 11/07/2022]
Abstract
Cell identity is largely determined by its transcriptional profile. In tumour, deregulation of transcription factor expression and/or activity enables cancer cell to acquire a stem-like state characterised by capacity to self-renew, differentiate and form tumours in vivo. These stem-like cancer cells are highly metastatic and therapy resistant, thus warranting a more complete understanding of the molecular mechanisms downstream of the transcription factors that mediate the establishment of stemness state. Here, we review recent research findings that provide a mechanistic link between the commonly deregulated transcription factors and stemness in cancer. In particular, we describe the role of master transcription factors (SOX, OCT4, NANOG, KLF, BRACHYURY, SALL, HOX, FOX and RUNX), signalling-regulated transcription factors (SMAD, β-catenin, YAP, TAZ, AP-1, NOTCH, STAT, GLI, ETS and NF-κB) and unclassified transcription factors (c-MYC, HIF, EMT transcription factors and P53) across diverse tumour types, thereby yielding a comprehensive overview identifying shared downstream targets, highlighting unique mechanisms and discussing complexities.
Collapse
Affiliation(s)
- Egle-Helene Ervin
- Botnar Research Centre, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, Old Road, Headington, Oxford, OX3 7LD, United Kingdom.
| | - Rhiannon French
- Botnar Research Centre, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, Old Road, Headington, Oxford, OX3 7LD, United Kingdom.
| | - Chao-Hui Chang
- Botnar Research Centre, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, Old Road, Headington, Oxford, OX3 7LD, United Kingdom.
| | - Siim Pauklin
- Botnar Research Centre, Nuffield Department of Orthopaedics, Rheumatology and Musculoskeletal Sciences, University of Oxford, Old Road, Headington, Oxford, OX3 7LD, United Kingdom.
| |
Collapse
|
|
82
|
Limbu KR, Chhetri RB, Oh YS, Baek DJ, Park EY. Mebendazole Impedes the Proliferation and Migration of Pancreatic Cancer Cells through SK1 Inhibition Dependent Pathway. MOLECULES (BASEL, SWITZERLAND) 2022; 27:molecules27238127. [PMID: 36500220 PMCID: PMC9739667 DOI: 10.3390/molecules27238127] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Figures] [Subscribe] [Scholar Register] [Received: 10/28/2022] [Revised: 11/16/2022] [Accepted: 11/21/2022] [Indexed: 11/25/2022]
Abstract
Pancreatic ductal adenocarcinoma (PDAC) has one of the highest mortality rates and requires the development of highly efficacious medications that can improve the efficiency of existing treatment methods. In particular, in PDAC, resistance to conventional chemotherapy reduces the effectiveness of anticancer drugs, decreasing the therapeutic efficiency. Sphingosine 1-phosphate (S1P), produced by sphingosine kinase (SK), plays a vital role in cancer growth, metastasis, chemotherapy, and drug resistance. Focusing on the structural characteristics of mebendazole (MBZ), we studied whether MBZ would affect metastasis, invasion, and drug resistance in cancer by lowering S1P production through inhibition of SK activity. MBZ selectively inhibited SK1 more than SK2 and regulated the levels of sphingolipids. MBZ inhibited the proliferation and migration of cancer cells in other PDAC cell lines. To determine whether the effect of MBZ on cancer cell growth and migration is S1P-mediated, S1P was treated, and the growth and migration of cancer cells were observed. It was found that MBZ inhibited S1P-induced cancer cell growth, and MBZ showed a growth inhibitory effect by regulating the JAK2/STAT3/Bcl-2 pathway. The phosphorylation of focal adhesion kinase (FAK), a transcription factor that regulates migration, was inhibited by MBZ, so it was found that the effect of MBZ regulates the migration of cancer cells through the S1P/FAK/vimentin pathway. In conclusion, our study suggests that the anthelmintic MBZ can be used as a potential therapeutic agent for treating PDAC and for structural synthesis studies of its analogs.
Collapse
Affiliation(s)
- Khem Raj Limbu
- College of Pharmacy, Mokpo National University, Mokpo 58554, Republic of Korea
| | | | - Yoon Sin Oh
- Department of Food and Nutrition, Eulji University, Seongnam 13135, Republic of Korea
| | - Dong Jae Baek
- College of Pharmacy, Mokpo National University, Mokpo 58554, Republic of Korea
- Correspondence: (D.J.B.); (E.-Y.P.)
| | - Eun-Young Park
- College of Pharmacy, Mokpo National University, Mokpo 58554, Republic of Korea
- Correspondence: (D.J.B.); (E.-Y.P.)
| |
Collapse
|
|
83
|
Bryl R, Piwocka O, Kawka E, Mozdziak P, Kempisty B, Knopik-Skrocka A. Cancer Stem Cells-The Insight into Non-Coding RNAs. Cells 2022; 11:cells11223699. [PMID: 36429127 PMCID: PMC9688207 DOI: 10.3390/cells11223699] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/18/2022] [Revised: 11/11/2022] [Accepted: 11/15/2022] [Indexed: 11/23/2022] Open
Abstract
Since their initial identification three decades ago, there has been extensive research regarding cancer stem cells (CSCs). It is important to consider the biology of cancer stem cells with a particular focus on their phenotypic and metabolic plasticity, the most important signaling pathways, and non-coding RNAs (ncRNAs) regulating these cellular entities. Furthermore, the current status of therapeutic approaches against CSCs is an important consideration regarding employing the technology to improve human health. Cancer stem cells have claimed to be one of the most important group of cells for the development of several common cancers as they dictate features, such as resistance to radio- and chemotherapy, metastasis, and secondary tumor formation. Therapies which could target these cells may develop into an effective strategy for tumor eradication and a hope for patients for whom this disease remains uncurable.
Collapse
Affiliation(s)
- Rut Bryl
- Section of Regenerative Medicine and Cancer Research, Natural Sciences Club, Faculty of Biology, Adam Mickiewicz University, 61-614 Poznań, Poland
| | - Oliwia Piwocka
- Section of Regenerative Medicine and Cancer Research, Natural Sciences Club, Faculty of Biology, Adam Mickiewicz University, 61-614 Poznań, Poland
- Department of Electroradiology, Poznan University of Medical Sciences, 61-701 Poznań, Poland
- Doctoral School, Poznan University of Medical Sciences, 61-701 Poznań, Poland
| | - Emilia Kawka
- Section of Regenerative Medicine and Cancer Research, Natural Sciences Club, Faculty of Biology, Adam Mickiewicz University, 61-614 Poznań, Poland
| | - Paul Mozdziak
- Prestage Department of Poultry Science, North Carolina State University, Raleigh, NC 27695, USA
| | - Bartosz Kempisty
- Prestage Department of Poultry Science, North Carolina State University, Raleigh, NC 27695, USA
- Department of Human Morphology and Embryology, Division of Anatomy, Medical University of Wrocław, 50-367 Wroclaw, Poland
- Department of Veterinary Surgery, Institute of Veterinary Medicine, Nicolaus Copernicus University, 87-100 Torun, Poland
- Correspondence: or
| | - Agnieszka Knopik-Skrocka
- Section of Regenerative Medicine and Cancer Research, Natural Sciences Club, Faculty of Biology, Adam Mickiewicz University, 61-614 Poznań, Poland
- Department of Cell Biology, Faculty of Biology, Adam Mickiewicz University, 61-614 Poznań, Poland
| |
Collapse
|
|
84
|
PTEN overexpression and nuclear β-catenin stabilization promote morular differentiation through induction of epithelial-mesenchymal transition and cancer stem cell-like properties in endometrial carcinoma. Cell Commun Signal 2022; 20:181. [PMID: 36411429 PMCID: PMC9677676 DOI: 10.1186/s12964-022-00999-w] [Citation(s) in RCA: 5] [Impact Index Per Article: 2.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 09/13/2022] [Accepted: 10/23/2022] [Indexed: 11/22/2022] Open
Abstract
BACKGROUND Although a lack of functional PTEN contributes to tumorigenesis in a wide spectrum of human malignancies, little is known about the functional role of its overexpression in the tumors. The current study focused on PTEN overexpression in endometrial carcinoma (Em Ca). METHODS The functional impact of PTEN overexpression was assessed by Em Ca cell lines. Immunohistochemical analyses were also conducted using 38 Em Ca with morular lesions. RESULTS Em Ca cell lines stably overexpressing PTEN (H6-PTEN) exhibited epithelial-mesenchymal transition (EMT)-like features, probably through β-catenin/Slug-meditated suppression of E-cadherin. PTEN overexpression also inhibited cell proliferation, accelerated cellular senescence, increased apoptotic features, and enhanced migration capability. Moreover, H6-PTEN cells exhibited cancer stem cell (CSC)-like properties, along with high expression of aldehyde dehydrogenase 1 and CD44s, a large ALDH 1high population, enriched spheroid formation, and β-catenin-mediated upregulation of cyclin D2, which is required for persistent CSC growth. In clinical samples, immunoreactivities for PTEN, as well as CSC-related molecules, were significantly higher in morular lesions as compared to the surrounding carcinomas. PTEN score was positively correlated with expression of nuclear β-catenin, cytoplasmic CD133, and CD44v6, and negatively with cell proliferation. Finally, estrogen receptor-α (ERα)-dependent expression of Ezrin-radixin-moesin-binding phophoprotein-50 (EBP50), a multifunctional scaffolding protein, acts as a negative regulator of morular formation by Em Ca cells through interacting with PTEN and β-catenin. CONCLUSION In the abscess of ERα/EBP50 expression, PTEN overexpression and nuclear β-catenin stabilization promote the establishment and maintenance of morular phenotype associated with EMT/CSC-like features in Em Ca cells. Video Abstract.
Collapse
|
|
85
|
LncRNA PTPRG-AS1 maintains stem-cell-like features and promotes oxaliplatin resistance in colorectal cancer via regulating the miR-665 and STAT3 axis. Mol Cell Toxicol 2022. [DOI: 10.1007/s13273-022-00314-1] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 11/18/2022]
|
|
86
|
Luan Y, Bao Y, Wang F. Artesunate regulates the proliferation and differentiation of neural stem cells by activating the JAK‑2/STAT‑3 signaling pathway in ischemic stroke. Exp Ther Med 2022; 25:2. [PMID: 36561626 PMCID: PMC9748661 DOI: 10.3892/etm.2022.11701] [Citation(s) in RCA: 3] [Impact Index Per Article: 1.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 02/13/2022] [Accepted: 07/04/2022] [Indexed: 11/17/2022] Open
Abstract
Ischemic stroke is one of the most common causes of disability and death globally; therefore, the repair and reconstruction of the central nervous system (CNS) after stroke is very important. Neural stem/progenitor cells (NSPCs) may be the key to cell replacement therapy to treat CNS damage. It has previously been reported that artesunate (ART) is involved in the regulation of the biological functions of NSPCs; however, the mechanism of action of ART remains unclear. In the present study, different concentrations of ART were used to treat NSPCs following oxygen-glucose deprivation (OGD). Cell viability and apoptosis were analyzed using Cell Counting Kit-8 assay and flow cytometry, respectively, whereas immunofluorescence analysis was used to measure the expression levels of the differentiation-related molecule doublecortin (DCX) and proliferating cell nuclear antigen (PCNA). Western blotting was performed to analyze the expression levels of molecules related to the JAK-2/STAT-3 signaling pathway. The present results indicated that treatment with ART following OGD significantly promoted the viability of NSPCs, inhibited the apoptosis of NSPCs, and promoted the expression of PCNA and DCX. Moreover, ART significantly downregulated the protein expression levels of phosphorylated (p)-JAK-2 and p-STAT-3. Furthermore, activation of the JAK-2/STAT-3 signaling pathway and treatment with ART reversed the effects of ART on the proliferation, apoptosis and differentiation of NSPCs. In conclusion, the present data suggested that ART may promote the proliferation and differentiation of NSPCs, and reduce the apoptosis of NSPCs, by inhibiting the JAK-2/STAT-3 signaling pathway. ART may potentially be used for the treatment of ischemic stroke.
Collapse
Affiliation(s)
- Yumin Luan
- Department of Rehabilitation Medicine, The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Yanan Bao
- Department of Thoracic Surgery, The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Fei Wang
- Department of Intensive Medicine, The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China,Correspondence to: Professor Fei Wang, Department of Intensive Medicine, The First Affiliated Hospital of Kunming Medical University, 295 Xichang Road, Kunming, Yunnan 650032, P.R. China
| |
Collapse
|
|
87
|
Chen T, Xu B, Chen H, Sun Y, Song J, Sun X, Zhang X, Hua W. Transcription factor NFE2L3 promotes the proliferation of esophageal squamous cell carcinoma cells and causes radiotherapy resistance by regulating IL-6. COMPUTER METHODS AND PROGRAMS IN BIOMEDICINE 2022; 226:107102. [PMID: 36108571 DOI: 10.1016/j.cmpb.2022.107102] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Subscribe] [Scholar Register] [Received: 05/16/2022] [Revised: 08/23/2022] [Accepted: 08/29/2022] [Indexed: 06/15/2023]
Abstract
OBJECTIVE To scrutinize the impact of overexpression and interference of NFE2L3 on radiosensitivity of esophageal squamous cell carcinoma cells (ESCC) and its downstream mechanism and to assess whether NFE2L3 expression alters in vivo radiosensitivity of ESCC by developing a subcutaneous tumor model in mice. METHODS Through RNA-Seq, we compared the differentially expressed genes between the ECA-109R cell line and its parent ECA-109 cell line. The differentially expressed genes were selected and verified by qRT-PCR. Transfection of ESCC cell lines with NFE2L3 inhibitor or mimic lentivirus constructs was done to study the activity of NFE2L3. To assess the effect of NFE2L3 on cellular growth and proliferation, clonogenic survival assay, EdU incorporation assay, and CCK-8 assay were done after irradiation. To probe how many irradiated DNA double-strand breaks were produced, the corresponding intensity of γ-H2AX foci were detected by immunofluorescence. Apoptotic cells were assayed by flow cytometry assay after irradiation; To investigate the downstream genes of NFE2L3, we knocked NFE2L3, and RNA-Seq was used to find out the downstream genes. qRT-PCR and western blot ensued to score associated protein profiles. The in vivo ESCC cell radiosensitivity was scrutinized by nude mouse xenograft models. RESULTS The differential genes between ECA-109R cells and its parent ECA-109 cells were compared by qRT-PCR to unveil a significant increase in NFE2L3 expression. Functional analysis indicated that NFE2L3 increased radioresistance in ESCC cells. Then, through high-throughput sequencing and bioinformatics analysis, IL-6 was found to be a hub gene that played a role downstream of NFE2L3 and was verified by qRT-PCR, western blot, and double luciferase reporter gene experiment. NFE2L3 could regulate ESCC cell radiosensitivity via the IL-6-STAT3 signaling pathway, and downregulation of IL-6 expression could reverse the effects of highly expressed NFE2L3. In vivo tumor xenograft experiments confirmed that NFE2L3 affects the sensitivity to radiation therapy. CONCLUSION NFE2L3 can affect the radiosensitivity of ESCC cells through IL-6 transcription and IL-6/STAT3 signaling pathway. This makes NFE2L3 a putative target to regulate ESCC cell radiosensitivity.
Collapse
Affiliation(s)
- Tingting Chen
- Department of Oncology, Clinical Medical College, Yangzhou University, Yangzhou, Jiangsu Province, PR China
| | - Bing Xu
- Department of Oncology, Clinical Medical College, Yangzhou University, Yangzhou, Jiangsu Province, PR China
| | - Hui Chen
- Department of Radiation Oncology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province, PR China
| | - Yuanyuan Sun
- Department of Radiation Oncology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province, PR China
| | - Jiahang Song
- Department of Radiation Oncology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province, PR China
| | - Xinchen Sun
- Department of Radiation Oncology, The First Affiliated Hospital of Nanjing Medical University, Nanjing, Jiangsu Province, PR China.
| | - Xizhi Zhang
- Department of Oncology, Clinical Medical College, Yangzhou University, Yangzhou, Jiangsu Province, PR China.
| | - Wei Hua
- Department of Oncology, Clinical Medical College, Yangzhou University, Yangzhou, Jiangsu Province, PR China.
| |
Collapse
|
|
88
|
NOP53 undergoes liquid-liquid phase separation and promotes tumor radio-resistance. Cell Death Dis 2022; 8:436. [PMCID: PMC9622906 DOI: 10.1038/s41420-022-01226-8] [Citation(s) in RCA: 6] [Impact Index Per Article: 3.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 08/09/2022] [Revised: 10/12/2022] [Accepted: 10/17/2022] [Indexed: 11/08/2022]
Abstract
Aberrant DNA damage response (DDR) axis remains the major molecular mechanism for tumor radio-resistance. We recently characterized liquid-liquid phase separation (LLPS) as an essential mechanism of DDR, and identified several key DDR factors as potential LLPS proteins, including nucleolar protein NOP53. In this study, we found that NOP53 formed highly concentrated droplets in vivo and in vitro, which had liquid-like properties including the fusion of adjacent condensates, rapid fluorescence recovery after photobleaching and the sensitivity to 1,6-hexanediol. Moreover, the intrinsically disordered region 1 (IDR1) is required for NOP53 phase separation. In addition, multivalent-arginine-rich linear motifs (M-R motifs), which are enriched in NOP53, were essential for its nucleolar localization, but were dispensable for the LLPS of NOP53. Functionally, NOP53 silencing diminished tumor cell growth, and significantly sensitized colorectal cancer (CRC) cells to radiotherapy. Mechanically, NOP53 negatively regulated p53 pathway in CRC cells treated with or without radiation. Importantly, data from clinical samples confirmed a correlation between NOP53 expression and tumor radio-resistance. Together, these results indicate an important role of NOP53 in radio-resistance, and provide a potential target for tumor radio-sensitization.
Collapse
|
|
89
|
Shao Y, Chen J, Hu Y, Wu Y, Zeng H, Lin S, Lai Q, Fan X, Zhou X, Zheng M, Gao B, Sun J. Investigating the effects and mechanisms of Erchen Decoction in the treatment of colorectal cancer by network pharmacology and experimental validation. Front Pharmacol 2022; 13:1000639. [PMID: 36313338 PMCID: PMC9606229 DOI: 10.3389/fphar.2022.1000639] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Grants] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/22/2022] [Accepted: 09/30/2022] [Indexed: 12/24/2022] Open
Abstract
Objective: Erchen Decoction (ECD), a well-known traditional Chinese medicine, exerts metabolism-regulatory, immunoregulation, and anti-tumor effects. However, the action and pharmacological mechanism of ECD remain largely unclear. In the present study, we explored the effects and mechanisms of ECD in the treatment of CRC using network pharmacology, molecular docking, and systematic experimental validation.Methods: The active components of ECD were obtained from the TCMSP database and the potential targets of them were annotated by the STRING database. The CRC-related targets were identified from different databases (OMIM, DisGeNet, GeneCards, and DrugBank). The interactive targets of ECD and CRC were screened and the protein-protein interaction (PPI) networks were constructed. Then, the hub interactive targets were calculated and visualized from the PPI network using the Cytoscape software. Gene Ontology (GO) and Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway enrichment analyses were performed. In addition, the molecular docking was performed. Finally, systematic in vitro, in vivo and molecular biology experiments were performed to further explore the anti-tumor effects and underlying mechanisms of ECD in CRC.Results: A total of 116 active components and 246 targets of ECD were predicted based on the component-target network analysis. 2406 CRC-related targets were obtained from different databases and 140 intersective targets were identified between ECD and CRC. 12 hub molecules (STAT3, JUN, MAPK3, TP53, MAPK1, RELA, FOS, ESR1, IL6, MAPK14, MYC, and CDKN1A) were finally screened from PPI network. GO and KEGG pathway enrichment analyses demonstrated that the biological discrepancy was mainly focused on the tumorigenesis-, immune-, and mechanism-related pathways. Based on the experimental validation, ECD could suppress the proliferation of CRC cells by inhibiting cell cycle and promoting cell apoptosis. In addition, ECD could inhibit tumor growth in mice. Finally, the results of molecular biology experiments suggested ECD could regulate the transcriptional levels of several hub molecules during the development of CRC, including MAPKs, PPARs, TP53, and STATs.Conclusion: This study revealed the potential pharmacodynamic material basis and underlying molecular mechanisms of ECD in the treatment of CRC, providing a novel insight for us to find more effective anti-CRC drugs.
Collapse
Affiliation(s)
- Yanfei Shao
- Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Shanghai Minimally Invasive Surgery Center, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
| | - Jingxian Chen
- Department of Traditional Chinese Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
| | - Yujie Hu
- College of Integrated Traditional Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou, China
| | - Yuan Wu
- Department of Traditional Chinese Medicine, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
| | - Hualin Zeng
- College of Integrated Traditional Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou, China
| | - Shuying Lin
- College of Integrated Traditional Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou, China
| | - Qiying Lai
- College of Integrated Traditional Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou, China
| | - Xiaodong Fan
- Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Shanghai Minimally Invasive Surgery Center, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
| | - Xueliang Zhou
- Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Shanghai Minimally Invasive Surgery Center, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
| | - Minhua Zheng
- Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Shanghai Minimally Invasive Surgery Center, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- *Correspondence: Jing Sun, ; Bizhen Gao, ; Minhua Zheng,
| | - Bizhen Gao
- College of Integrated Traditional Chinese and Western Medicine, Fujian University of Traditional Chinese Medicine, Fuzhou, China
- *Correspondence: Jing Sun, ; Bizhen Gao, ; Minhua Zheng,
| | - Jing Sun
- Department of General Surgery, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- Shanghai Minimally Invasive Surgery Center, Ruijin Hospital, Shanghai Jiao Tong University School of Medicine, Shanghai, China
- *Correspondence: Jing Sun, ; Bizhen Gao, ; Minhua Zheng,
| |
Collapse
|
|
90
|
Manni W, Min W. Signaling pathways in the regulation of cancer stem cells and associated targeted therapy. MedComm (Beijing) 2022; 3:e176. [PMID: 36226253 PMCID: PMC9534377 DOI: 10.1002/mco2.176] [Citation(s) in RCA: 14] [Impact Index Per Article: 7.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/11/2022] [Revised: 08/20/2022] [Accepted: 08/22/2022] [Indexed: 11/07/2022] Open
Abstract
Cancer stem cells (CSCs) are defined as a subpopulation of malignant tumor cells with selective capacities for tumor initiation, self-renewal, metastasis, and unlimited growth into bulks, which are believed as a major cause of progressive tumor phenotypes, including recurrence, metastasis, and treatment failure. A number of signaling pathways are involved in the maintenance of stem cell properties and survival of CSCs, including well-established intrinsic pathways, such as the Notch, Wnt, and Hedgehog signaling, and extrinsic pathways, such as the vascular microenvironment and tumor-associated immune cells. There is also intricate crosstalk between these signal cascades and other oncogenic pathways. Thus, targeting pathway molecules that regulate CSCs provides a new option for the treatment of therapy-resistant or -refractory tumors. These treatments include small molecule inhibitors, monoclonal antibodies that target key signaling in CSCs, as well as CSC-directed immunotherapies that harness the immune systems to target CSCs. This review aims to provide an overview of the regulating networks and their immune interactions involved in CSC development. We also address the update on the development of CSC-directed therapeutics, with a special focus on those with application approval or under clinical evaluation.
Collapse
Affiliation(s)
- Wang Manni
- Department of Biotherapy, Cancer Center, West China HospitalSichuan UniversityChengduP. R. China
| | - Wu Min
- Department of Biomedical Sciences, School of Medicine and Health SciencesUniversity of North DakotaGrand ForksNorth DakotaUSA
| |
Collapse
|
|
91
|
Si Y, Li L, Zhang W, Liu Q, Liu B. GANT61 exerts anticancer cell and anticancer stem cell capacity in colorectal cancer by blocking the Wnt/β‑catenin and Notch signalling pathways. Oncol Rep 2022; 48:182. [PMID: 36069229 PMCID: PMC9478957 DOI: 10.3892/or.2022.8397] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 06/28/2020] [Accepted: 05/13/2021] [Indexed: 12/24/2022] Open
Abstract
The present study aimed to assess the anticancer cell and anticancer stem cell (CSC) effects of GANT61, and its regulatory influence on the Wnt/β-catenin and Notch signalling pathways in colorectal cancer (CRC). HT-29 and HCT-116 cells were treated with 0, 2.5, 5, 10, 20 or 40 µM GANT61, after which relative cell viability and the expression of Gli1, β-catenin and Notch1, as well as the percentage of CD133+ cells, were detected. Subsequently, HT-29/HCT-116 cells and CSCs were treated with 20 µM GANT61, 10 mM of the Wnt/β-catenin pathway agonist HLY78, and 30 mM of the Notch pathway agonist JAG1 (alone or in combination), which was followed by the assessment of cell viability and apoptosis. In both cell lines, GANT61 reduced relative cell viability in a time- and dose-dependent manner, inhibited Gli1, β-catenin and Notch1 expression, and decreased the percentage of CD133+ cells in a dose-dependent manner. Furthermore, HLY78 and JAG1 were both found to improve the relative viability, while downregulating the apoptosis of untreated and GANT61-treated HT-29 and HCT-116 cells. Moreover, Wnt/β-catenin and Notch signalling pathway activity were upregulated in CSCs isolated from HT-29 and HCT-116 cells, compared with the associated control groups. GANT61 also reduced the viability of HT-29 and HCT-116 cells and increased apoptosis, whereas HLY78 and JAG1 treatment resulted in the opposite effect. Moreover, both HLY78 and JAG1 attenuated the effects of GANT61 on cellular viability and apoptosis. In conclusion, GANT61 was found to effectively eliminate cancer cells and CSCs by blocking the Wnt/β-catenin and Notch signalling pathways in CRC.
Collapse
Affiliation(s)
- Yanhui Si
- Department of General Surgery, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, P.R. China
| | - Lei Li
- Department of General Surgery, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, P.R. China
| | - Weiwei Zhang
- Department of General Surgery, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, P.R. China
| | - Qiling Liu
- Department of General Surgery, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, P.R. China
| | - Baochi Liu
- Department of General Surgery, Shanghai Public Health Clinical Center, Fudan University, Shanghai 201508, P.R. China
| |
Collapse
|
|
92
|
E3 Ubiquitin Ligase CHIP Inhibits the Interaction between Hsp90β and MAST1 to Repress Radiation Resistance in Non-Small-Cell Lung Cancer Stem Cells. Stem Cells Int 2022; 2022:2760899. [PMID: 36199626 PMCID: PMC9527118 DOI: 10.1155/2022/2760899] [Citation(s) in RCA: 4] [Impact Index Per Article: 2.0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 03/28/2022] [Revised: 08/12/2022] [Accepted: 08/18/2022] [Indexed: 11/24/2022] Open
Abstract
The radiation resistance of cancer stem cells poses a critical obstacle for management of non-small-cell lung cancer (NSCLC). It is interesting to note that E3 ubiquitin ligase CHIP is involved in radiation resistance and stemness phenotypes in NSCLC, while the downstream mechanisms remain elusive. Therefore, this study is aimed at exploring the possible molecular mechanism of E3 ubiquitin ligase CHIP in radiation resistance of NSCLC stem cells. Cancer and adjacent normal tissues of NSCLC patients were collected to determine expression of CHIP, Hsp90β, and MAST1. CD133+ cells were isolated from the NSCLC tissues and the lung cancer cell line A549 by flow cytometric sorting. Accordingly, downregulated CHIP and upregulated Hsp90β and MAST1 were observed in cancer tissues from NSCLC patients and in NSCLC stem cells. Sphere formation assay, colony formation assay, and flow cytometry were performed to examine self-renewal ability, survival, and apoptosis of NSCLC stem cells. An animal model of tumor xenograft was developed in nude mice to observe the tumorigenic ability and radiation resistance of NSCLC stem cells. CHIP overexpression was demonstrated to inhibit the NSCLC stem cell properties and radiation resistance in vitro and in vivo. Mechanistically, CHIP promoted MAST1 ubiquitination by blocking Hsp90β interaction with MAST1, thus inhibiting MAST1 protein stability. Furthermore, CHIP-mediated downregulation of MAST1 protein stability inhibited the NSCLC stem cell properties and radiation resistance. Collectively, CHIP promotes the ubiquitination of MAST1 by blocking the interaction of Hsp90β with MAST1, leading to decreased MAST1 protein stability, which suppressed NSCLC stem cell properties and radiation resistance.
Collapse
|
|
93
|
DiNatale A, Castelli MS, Nash B, Meucci O, Fatatis A. Regulation of Tumor and Metastasis Initiation by Chemokine Receptors. J Cancer 2022; 13:3160-3176. [PMID: 36118530 PMCID: PMC9475358 DOI: 10.7150/jca.72331] [Citation(s) in RCA: 1] [Impact Index Per Article: 0.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 02/23/2022] [Accepted: 08/10/2022] [Indexed: 12/13/2022] Open
Abstract
Tumor-initiating cells (TICs) are a rare sub-population of cells within the bulk of a tumor that are major contributors to tumor initiation, metastasis, and chemoresistance. TICs have a stem-cell-like phenotype that is dictated by the expression of master regulator transcription factors, including OCT4, NANOG, and SOX2. These transcription factors are expressed via activation of multiple signaling pathways that drive cancer initiation and progression. Importantly, these same signaling pathways can be activated by select chemokine receptors. Chemokine receptors are increasingly being revealed as major drivers of the TIC phenotype, as their signaling can lead to activation of stemness-controlling transcription factors. Additionally, the cell surface expression of chemokine receptors provides a unique therapeutic target to disrupt signaling pathways that control the expression of master regulator transcription factors and the TIC phenotype. This review summarizes the master regulator transcription factors known to dictate the TIC phenotype, along with the complex signaling pathways that can mediate their expression and the chemokine receptors that are most upstream of this phenotype.
Collapse
Affiliation(s)
- Anthony DiNatale
- Department of Pharmacology and Physiology, Drexel University College of Medicine, Philadelphia, PA 19102, USA.,Present Address: Janssen Oncology, Spring House, PA, USA
| | - Maria Sofia Castelli
- Department of Pharmacology and Physiology, Drexel University College of Medicine, Philadelphia, PA 19102, USA.,Present address: Perelman School of Medicine, University of Pennsylvania, Philadelphia, PA 19104, USA
| | - Bradley Nash
- Department of Pharmacology and Physiology, Drexel University College of Medicine, Philadelphia, PA 19102, USA
| | - Olimpia Meucci
- Department of Pharmacology and Physiology, Drexel University College of Medicine, Philadelphia, PA 19102, USA.,Program in Immune Cell Regulation & Targeting, Sidney Kimmel Cancer Center of Thomas Jefferson University, Philadelphia, PA 19107, USA
| | - Alessandro Fatatis
- Department of Pharmacology and Physiology, Drexel University College of Medicine, Philadelphia, PA 19102, USA.,Program in Translational and Cellular Oncology, Sidney Kimmel Cancer Center of Thomas Jefferson University, Philadelphia, PA 19107, USA
| |
Collapse
|
|
94
|
Mahapatra S, Sharma MVR, Brownson B, Gallicano VE, Gallicano GI. Cardiac inducing colonies halt fibroblast activation and induce cardiac/endothelial cells to move and expand via paracrine signaling. Mol Biol Cell 2022; 33:ar96. [PMID: 35653297 DOI: 10.1091/mbc.e22-02-0032] [Citation(s) in RCA: 2] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 11/11/2022] Open
Abstract
Myocardial fibrosis (MF), a common event that develops after myocardial infarction, initially is a reparative process but eventually leads to heart failure and sudden cardiac arrest. In MF, the infarct area is replaced by a collagenous-based scar induced by "excessive" collagen deposition from activated cardiac fibroblasts. The scar prevents ventricular wall thinning; however, over time it expands to noninfarcted myocardium. Therapies to prevent fibrosis include reperfusion, anti-fibrotic agents, and ACE inhibitors. Paracrine factor (PF)/stem cell research has recently gained significance as a therapy. We consistently find that cardiac inducing colonies (CiCs) (derived from human germline pluripotent stem cells) secrete PFs at physiologically relevant concentrations that suppress cardiac fibroblast activation and excessive extracellular matrix protein secretion. These factors also affect human cardiomyocytes and endothelial cells by inducing migration/proliferation of both populations into a myocardial wound model. Finally, CiC factors modulate matrix turnover and proinflammation. Taking the results together, we show that CiCs could help tip the balance from fibrosis toward repair.
Collapse
Affiliation(s)
- Samiksha Mahapatra
- Department of Biochemistry and Molecular Biology, Georgetown University Medical Center, Washington, DC 20057-145
| | | | - Breanna Brownson
- Department of Biochemistry and Molecular Biology, Georgetown University Medical Center, Washington, DC 20057-145.,Rye High School, Rye, NY 10580
| | - Vaughn E Gallicano
- Department of Biochemistry and Molecular Biology, Georgetown University Medical Center, Washington, DC 20057-145.,Thomas Edison High School, Alexandria, VA 22310
| | - G Ian Gallicano
- Department of Biochemistry and Molecular Biology, Georgetown University Medical Center, Washington, DC 20057-145
| |
Collapse
|
|
95
|
Huang L, Gong J, Hu Y, Tan QL, Liu B, Yu XW, Hao XL, Guo QN. Long-term exposure to low levels of okadaic acid accelerates cell cycle progression in colonic epithelial cells via p53 and Jak/Stat3 signaling pathways. Heliyon 2022; 8:e10444. [PMID: 36105456 PMCID: PMC9465354 DOI: 10.1016/j.heliyon.2022.e10444] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/19/2022] [Revised: 07/04/2022] [Accepted: 08/22/2022] [Indexed: 11/30/2022] Open
Abstract
As a major component of diarrheic shellfish poisoning (DSP) toxins, okadaic acid (OA) is widely distributed worldwide, and causes a series of serious public health problems. In colon tissue, previous studies have shown that high doses of OA can affect various intracellular processes, including destroy intercellular communication at gap junctions, induce cell apoptosis and trigger cell cycle arrest. However, there is a scarcity of studies on the effect and mechanism of action of low doses of OA in colonic tissues. In this study, we observed that exposure to low levels of OA altered cell cycle progression in vitro and in vivo. Investigation of the underlying mechanism revealed that OA induced alterations in the cell cycle by inhibiting the p53 signaling pathway or inducing the Jak/Stat3 signaling pathway. In conclusion, this study provides novel insights into the effect and mechanism underlying long-term exposure to low levels of OA. Long-term exposure to low levels of OA accelerates cell cycles in vitro and in vivo OA induced changes in cell cycle by inhibiting the p53 signaling pathway OA induced changes in cell cycle by inducing the Jak/Stat3 signaling pathway
Collapse
|
|
96
|
Lu Z, Zheng X, Ding C, Zou Z, Liang Y, Zhou Y, Li X. Deciphering the Biological Effects of Radiotherapy in Cancer Cells. Biomolecules 2022; 12:biom12091167. [PMID: 36139006 PMCID: PMC9496570 DOI: 10.3390/biom12091167] [Citation(s) in RCA: 10] [Impact Index Per Article: 5.0] [Reference Citation Analysis] [Abstract] [MESH Headings] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/22/2022] [Revised: 08/15/2022] [Accepted: 08/19/2022] [Indexed: 12/12/2022] Open
Abstract
Radiotherapy remains an effective conventional method of treatment for patients with cancer. However, the clinical efficacy of radiotherapy is compromised by the development of radioresistance of the tumor cells during the treatment. Consequently, there is need for a comprehensive understanding of the regulatory mechanisms of tumor cells in response to radiation to improve radiotherapy efficacy. The current study aims to highlight new developments that illustrate various forms of cancer cell death after exposure to radiation. A summary of the cellular pathways and important target proteins that are responsible for tumor radioresistance and metastasis is also provided. Further, the study outlines several mechanistic descriptions of the interaction between ionizing radiation and the host immune system. Therefore, the current review provides a reference for future research studies on the biological effects of new radiotherapy technologies, such as ultra-high-dose-rate (FLASH) radiotherapy, proton therapy, and heavy-ion therapy.
Collapse
Affiliation(s)
| | | | | | | | | | - Yan Zhou
- Correspondence: (Y.Z.); (X.L.); Tel.: +86-0816-225-2295 (Y.Z.); +86-0816-220-6272 (X.L.)
| | - Xiaoan Li
- Correspondence: (Y.Z.); (X.L.); Tel.: +86-0816-225-2295 (Y.Z.); +86-0816-220-6272 (X.L.)
| |
Collapse
|
|
97
|
Zafari N, Khosravi F, Rezaee Z, Esfandyari S, Bahiraei M, Bahramy A, Ferns GA, Avan A. The role of the tumor microenvironment in colorectal cancer and the potential therapeutic approaches. J Clin Lab Anal 2022; 36:e24585. [PMID: 35808903 PMCID: PMC9396196 DOI: 10.1002/jcla.24585] [Citation(s) in RCA: 9] [Impact Index Per Article: 4.5] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 03/15/2022] [Revised: 06/07/2022] [Accepted: 06/23/2022] [Indexed: 11/26/2022] Open
Abstract
BACKGROUND Colorectal cancer (CRC) with a high prevalence is recognized as the fourth most common cause of cancer-related death globally. Over the past decade, there has been growing interest in the network of tumor cells, stromal cells, immune cells, blood vessel cells, and fibroblasts that comprise the tumor microenvironment (TME) to identify new therapeutic interventions. METHODS Databases, such as Google Scholar, PubMed, and Scopus, were searched to provide an overview of the recent research progress related to targeting the TME as a novel therapeutic approach. RESULTS Tumor microenvironment as a result of the cross talk between these cells may result in either advantages or disadvantages in tumor development and metastasis, affecting the signals and responses from the surrounding cells. Whilst chemotherapy has led to an improvement in CRC patients' survival, the metastatic aspect of the disease remains difficult to avoid. CONCLUSIONS The present review emphasizes the structure and function of the TME, alterations in the TME, its role in the incidence and progression of CRC, the effects on tumor development and metastasis, and also the potential of its alterations as therapeutic targets. It should be noted that providing novel studies in this field of research might help us to achieve practical therapeutic strategies based on their interaction.
Collapse
Affiliation(s)
- Narges Zafari
- Department of Medical Genetics, School of MedicineTehran University of Medical SciencesTehranIran
| | - Fatemeh Khosravi
- Molecular Medicine Research Center, Hormozgan Health InstituteHormozgan University of Medical SciencesBandar AbbasIran
| | - Zahra Rezaee
- Department of Medical Genetics, Faculty of Medical SciencesTarbiat Modares UniversityTehranIran
| | - Sahar Esfandyari
- Department of Anatomy, School of MedicineTehran University of Medical SciencesTehranIran
| | - Mohamad Bahiraei
- Department of Radiology, Besat HospitalHamedan University of Medical SciencesHamedanIran
| | - Afshin Bahramy
- Department of Medical Genetics, Faculty of Medical SciencesTarbiat Modares UniversityTehranIran
| | - Gordon A. Ferns
- Brighton & Sussex Medical SchoolDivision of Medical EducationSussexUK
| | - Amir Avan
- Metabolic Syndrome Research CenterMashhad University of Medical SciencesMashhadIran
- Basic Medical Sciences InstituteMashhad University of Medical SciencesMashhadIran
- Medical Genetics Research CenterMashhad University of Medical SciencesMashhadIran
| |
Collapse
|
|
98
|
Properties of Leukemic Stem Cells in Regulating Drug Resistance in Acute and Chronic Myeloid Leukemias. Biomedicines 2022; 10:biomedicines10081841. [PMID: 36009388 PMCID: PMC9405586 DOI: 10.3390/biomedicines10081841] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/04/2022] [Revised: 07/26/2022] [Accepted: 07/27/2022] [Indexed: 11/17/2022] Open
Abstract
Notoriously known for their capacity to reconstitute hematological malignancies in vivo, leukemic stem cells (LSCs) represent key drivers of therapeutic resistance and disease relapse, posing as a major medical dilemma. Despite having low abundance in the bulk leukemic population, LSCs have developed unique molecular dependencies and intricate signaling networks to enable self-renewal, quiescence, and drug resistance. To illustrate the multi-dimensional landscape of LSC-mediated leukemogenesis, in this review, we present phenotypical characteristics of LSCs, address the LSC-associated leukemic stromal microenvironment, highlight molecular aberrations that occur in the transcriptome, epigenome, proteome, and metabolome of LSCs, and showcase promising novel therapeutic strategies that potentially target the molecular vulnerabilities of LSCs.
Collapse
|
|
99
|
Hajimoradi M, Rezalotfi A, Esmaeilnejad-Ahranjani P, Mohammad Hassan Z, Ebrahimi M. STAT3 inactivation suppresses stemness properties in gastric cancer stem cells and promotes Th17 in Treg/Th17 balance. Int Immunopharmacol 2022; 111:109048. [PMID: 35905563 DOI: 10.1016/j.intimp.2022.109048] [Citation(s) in RCA: 8] [Impact Index Per Article: 4.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/01/2022] [Revised: 07/07/2022] [Accepted: 07/08/2022] [Indexed: 11/05/2022]
Abstract
Signal transducer and activator of transcription 3 (STAT3) has been recognized with dual effects in provision of cancer; either tumor inductive or immune suppressive. Recent findings considering the role of STAT3 in stem cells and cancer stem cell regulation, but its role in gastric cancer stem cells (GCSCs) and modulating the Th17/Treg balance is unknown. In the present study, we aimed to evaluate the role of activated STAT3 in GCSCs and Th17/ Treg cell paradigm. In completion of our previous results, the findings here indicate that gastro-spheroids, as a model of GCSCs, represent higher level of STAT3 activity, up-regulation of TGF-b and VEGF with downregulation of IL-6. On the other hand, treatment of normal naïve T cells with conditioned medium derived from gastro-spheroids promotes T cell differentiation toward cells with a higher level of FOXP3, TGF-b, and IL-10 expression which is indicative of Treg cells. Suppression of STAT3 activation in cancer cells by using Stattic small molecule treatment, decreases stemness features (i.e. spheroid formation and integrity, stemness gene expression and in vivo tumorigenicity capacity) and downregulates TGF-b in the cancer cells. Furthermore, co-culture of conditioned medium of STAT3 inhibited cancer cells with normal PBMCs leads to reduction in the percentage of Treg accompanied with increase of Th17 cells with a decrease in the secretion of TGF-b and increase in IFN-γ in T cells under differentiation. Therefore, targeting the STAT3 pathway in cancer cells seems to control the tumor formation and also impact on immune cells shifting to antitumor Th17 population.
Collapse
Affiliation(s)
- Monireh Hajimoradi
- Department of Immunology, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran; Department of Stem Cells and Developmental Biology, Cell Sciences Research Center, Royan Institute for Stem Cell Biology and Technology, ACECR, Tehran, Iran; Department of Immunology, Razi Vaccine and Serum Research Institute, Agricultural Research, Education and Extension Organization (AREEO), Karaj, Iran
| | - Alaleh Rezalotfi
- Department of Stem Cells and Developmental Biology, Cell Sciences Research Center, Royan Institute for Stem Cell Biology and Technology, ACECR, Tehran, Iran; Institute of Immunology, Hannover Medical School, 30625 Hannover, Germany
| | - Parvaneh Esmaeilnejad-Ahranjani
- Department of Anaerobic Bacterial Vaccine Research and Production, Razi Vaccine and Serum Research Institute, Agricultural Research, Education and Extension Organization (AREEO), Karaj, Iran
| | - Zuhair Mohammad Hassan
- Department of Immunology, Faculty of Medical Sciences, Tarbiat Modares University, Tehran, Iran.
| | - Marzieh Ebrahimi
- Department of Stem Cells and Developmental Biology, Cell Sciences Research Center, Royan Institute for Stem Cell Biology and Technology, ACECR, Tehran, Iran.
| |
Collapse
|
|
100
|
Zhang F, Li W, Cui Q, Chen Y, Liu Y. Case Report: Immune Microenvironment and Mutation Features in a Patient With Epstein–Barr Virus Positive Large B-Cell Lymphoma Secondary to Angioimmunoblastic T-Cell Lymphoma. Front Genet 2022; 13:940513. [PMID: 35938041 PMCID: PMC9354849 DOI: 10.3389/fgene.2022.940513] [Citation(s) in RCA: 1] [Impact Index Per Article: 0.5] [Reference Citation Analysis] [Abstract] [Grants] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/10/2022] [Accepted: 06/20/2022] [Indexed: 01/24/2023] Open
Abstract
On rare occasions, secondary Epstein–Barr virus (EBV)-associated B-cell lymphoma can develop in patients with angioimmunoblastic T-cell lymphoma (AITL). Here, we describe the tumor microenvironment and mutation features of a patient with EBV + large B-cell lymphoma (LBCL) secondary to AITL. He was admitted to hospital due to a 1-year history of fever and enlarged right inguinal lymph nodes. A biopsy of the right inguinal lymph node demonstrated that numerous diffuse medium-sized atypical lymphocytes proliferated, together with increased extrafollicular follicular dendritic cell meshwork, and the lymphocytes expressed CD3, CD4, BCL6, CD10, PD-1, CXCL13, and Ki-67 (75%). Thus, a diagnosis of AITL was made. However, the disease progressed following treatment by CHOP regimen (cyclophosphamide, adriamycin, vincristine, and prednisone). Biopsy showed that most of the cells were positive for CD20 staining and IgH rearrangement. Analysis of 22 kinds of immune cells showed that the numbers of activated NK cells and activated memory T cells increased, while the T-follicular helper population decreased in the transformed sample. In addition, compared with the primary sample, RHOA (G17V) mutation was not detected, while JAK2 and TRIP12 gene mutations were detected in the transformed sample. Overall, we described the immune microenvironment and mutation features of a patient with EBV + LBCL secondary to AITL. This study will help us to understand the mechanisms by which AITL transforms to B-cell lymphoma.
Collapse
|