|
1
|
Pedroza-Torres A, Romero-Córdoba SL, Montaño S, Peralta-Zaragoza O, Vélez-Uriza DE, Arriaga-Canon C, Guajardo-Barreto X, Bautista-Sánchez D, Sosa-León R, Hernández-González O, Díaz-Chávez J, Alvarez-Gómez RM, Herrera LA. Radio-miRs: a comprehensive view of radioresistance-related microRNAs. Genetics 2024; 227:iyae097. [PMID: 38963803 PMCID: PMC11304977 DOI: 10.1093/genetics/iyae097] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/02/2024] [Accepted: 05/29/2024] [Indexed: 07/06/2024] Open
Abstract
Radiotherapy is a key treatment option for a wide variety of human tumors, employed either alone or alongside with other therapeutic interventions. Radiotherapy uses high-energy particles to destroy tumor cells, blocking their ability to divide and proliferate. The effectiveness of radiotherapy is due to genetic and epigenetic factors that determine how tumor cells respond to ionizing radiation. These factors contribute to the establishment of resistance to radiotherapy, which increases the risk of poor clinical prognosis of patients. Although the mechanisms by which tumor cells induce radioresistance are unclear, evidence points out several contributing factors including the overexpression of DNA repair systems, increased levels of reactive oxygen species, alterations in the tumor microenvironment, and enrichment of cancer stem cell populations. In this context, dysregulation of microRNAs or miRNAs, critical regulators of gene expression, may influence how tumors respond to radiation. There is increasing evidence that miRNAs may act as sensitizers or enhancers of radioresistance, regulating key processes such as the DNA damage response and the cell death signaling pathway. Furthermore, expression and activity of miRNAs have shown informative value in overcoming radiotherapy and long-term radiotoxicity, revealing their potential as biomarkers. In this review, we will discuss the molecular mechanisms associated with the response to radiotherapy and highlight the central role of miRNAs in regulating the molecular mechanisms responsible for cellular radioresistance. We will also review radio-miRs, radiotherapy-related miRNAs, either as sensitizers or enhancers of radioresistance that hold promise as biomarkers or pharmacological targets to sensitize radioresistant cells.
Collapse
Affiliation(s)
- Abraham Pedroza-Torres
- Programa Investigadoras e Investigadores por México, Consejo Nacional de Humanidades, Ciencias y Tecnologías, Mexico City C.P. 03940, Mexico
- Clínica de Cáncer Hereditario, Instituto Nacional de Cancerología, Mexico City C.P. 14080, Mexico
| | - Sandra L Romero-Córdoba
- Departamento de Medicina Genómica y Toxicología Ambiental, Instituto de Investigaciones Biomédicas, Universidad Nacional Autónoma de México, Mexico City C.P. 04510, Mexico
- Departamento de Bioquímica, Instituto Nacional de Ciencias Médicas y Nutrición “Salvador Zubirán”, Mexico City C.P. 14080, Mexico
| | - Sarita Montaño
- Laboratorio de Bioinformática, Facultad de Ciencias Químico-Biológicas, Universidad Autónoma de Sinaloa (FCQB-UAS), Culiacán Rosales, Sinaloa C.P. 80030, Mexico
| | - Oscar Peralta-Zaragoza
- Dirección de Infecciones Crónicas y Cáncer, Centro de Investigación Sobre Enfermedades Infecciosas, Instituto Nacional de Salud Pública, Cuernavaca, Morelos C.P. 62100, Mexico
| | - Dora Emma Vélez-Uriza
- Laboratorio de Traducción y Cáncer, Instituto Nacional de Cancerología, Mexico City C.P. 14080, Mexico
| | - Cristian Arriaga-Canon
- Unidad de Investigación Biomédica en Cáncer, Instituto Nacional de Cancerología–Instituto de Investigaciones Biomédicas–Universidad Nacional Autónoma de México (UNAM), Mexico City C.P. 14080, Mexico
- Tecnológico de Monterrey, Escuela de Medicina y Ciencias de la Salud, Monterrey, Nuevo León C.P. 64710, Mexico
| | - Xiadani Guajardo-Barreto
- Unidad de Investigación Biomédica en Cáncer, Instituto Nacional de Cancerología–Instituto de Investigaciones Biomédicas–Universidad Nacional Autónoma de México (UNAM), Mexico City C.P. 14080, Mexico
| | - Diana Bautista-Sánchez
- Department of Microbiology and Immunology, Life Sciences Institute, University of British Columbia, Vancouver, BC V6T 1Z3, Canada
| | - Rodrigo Sosa-León
- Clínica de Cáncer Hereditario, Instituto Nacional de Cancerología, Mexico City C.P. 14080, Mexico
| | - Olivia Hernández-González
- Laboratorio de Microscopia Electrónica, Instituto Nacional de Rehabilitación “Luis Guillermo Ibarraa Ibarra”, Mexico City C.P. 14389, Mexico
| | - José Díaz-Chávez
- Unidad de Investigación Biomédica en Cáncer, Instituto Nacional de Cancerología–Instituto de Investigaciones Biomédicas–Universidad Nacional Autónoma de México (UNAM), Mexico City C.P. 14080, Mexico
| | - Rosa María Alvarez-Gómez
- Clínica de Cáncer Hereditario, Instituto Nacional de Cancerología, Mexico City C.P. 14080, Mexico
| | - Luis A Herrera
- Unidad de Investigación Biomédica en Cáncer, Instituto Nacional de Cancerología–Instituto de Investigaciones Biomédicas–Universidad Nacional Autónoma de México (UNAM), Mexico City C.P. 14080, Mexico
- Tecnológico de Monterrey, Escuela de Medicina y Ciencias de la Salud, Monterrey, Nuevo León C.P. 64710, Mexico
| |
Collapse
|
|
2
|
Guo L, Ding G, Ba Y, Tan B, Tian L, Wang K. Transcription factor STAT4 counteracts radiotherapy resistance in breast carcinoma cells by activating the MALAT1/miR-21-5p/THRB regulatory network. Am J Cancer Res 2024; 14:1501-1522. [PMID: 38726265 PMCID: PMC11076251 DOI: 10.62347/vsju7227] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/23/2024] [Accepted: 03/31/2024] [Indexed: 05/12/2024] Open
Abstract
Considering the limited research and the prevailing evidence of STAT4's tumor-suppressing role in breast carcinoma (BC) or in breast radiotherapy (RT) sensitivity requires more in-depth exploration. Our study delves into how STAT4, a transcription factor, affects BC cell resistance to radiotherapy by regulating the MALAT1/miR-21-5p/THRB axis. Bioinformatics analysis was performed to predict the regulatory mechanisms associated with STAT4 in BC. Subsequently, we identified the expression profiles of STAT4, MALAT1, miR-21-5p, and THRB in various tissues and cell lines, exploring their interactions and impact on RT resistance in BC cells. Moreover, animal models were established with X-ray irradiation for further validation. We discovered that STAT4, which is found to be minimally expressed in breast carcinoma (BC) tissues and cell lines, has been associated with a poorer prognosis. In vitro cellular assays indicated that STAT4 could mitigate radiotherapy resistance in BC cells by transcriptional activation of MALAT1. Additionally, MALAT1 up-regulated THRB expression by adsorbing miR-21-5p. As demonstrated in vitro and in vivo, overexpressing STAT4 inhibited miR-21-5p and enhanced THRB levels through transcriptional activation of MALAT1, which ultimately contributes to the reversal of radiotherapy resistance in BC cells and the suppression of tumor formation in nude mice. Collectively, STAT4 could inhibit miR-21-5p and up-regulate THRB expression through transcriptional activation of MALAT1, thereby mitigating BC cell resistance to radiotherapy and ultimately preventing BC development and progression.
Collapse
Affiliation(s)
| | | | - Yuntao Ba
- Department of Radiation, The Affiliated Cancer Hospital of Zhengzhou University & Henan Cancer HospitalZhengzhou 450008, Henan, China
| | - Bo Tan
- Department of Radiation, The Affiliated Cancer Hospital of Zhengzhou University & Henan Cancer HospitalZhengzhou 450008, Henan, China
| | - Lingling Tian
- Department of Radiation, The Affiliated Cancer Hospital of Zhengzhou University & Henan Cancer HospitalZhengzhou 450008, Henan, China
| | - Kunlun Wang
- Department of Radiation, The Affiliated Cancer Hospital of Zhengzhou University & Henan Cancer HospitalZhengzhou 450008, Henan, China
| |
Collapse
|
|
3
|
Seo Y, Kim DK, Park J, Park SJ, Park JJ, Cheon JH, Kim TI. A Comprehensive Understanding of Post-Translational Modification of Sox2 via Acetylation and O-GlcNAcylation in Colorectal Cancer. Cancers (Basel) 2024; 16:1035. [PMID: 38473392 DOI: 10.3390/cancers16051035] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/31/2024] [Revised: 02/24/2024] [Accepted: 03/01/2024] [Indexed: 03/14/2024] Open
Abstract
Aberrant expression of the pluripotency-associated transcription factor Sox2 is associated with poor prognosis in colorectal cancer (CRC). We investigated the regulatory roles of major post-translational modifications in Sox2 using two CRC cell lines, SW480 and SW620, derived from the same patient but with low and high Sox2 expression, respectively. Acetylation of K75 in the Sox2 nuclear export signal was relatively increased in SW480 cells and promotes Sox2 nucleocytoplasmic shuttling and proteasomal degradation of Sox2. LC-MS-based proteomics analysis identified HDAC4 and p300 as binding partners involved in the acetylation-mediated control of Sox2 expression in the nucleus. Sox2 K75 acetylation is mediated by the acetyltransferase activity of CBP/p300 and ACSS3. In SW620 cells, HDAC4 deacetylates K75 and is regulated by miR29a. O-GlcNAcylation on S246, in addition to K75 acetylation, also regulates Sox2 stability. These findings provide insights into the regulation of Sox2 through multiple post-translational modifications and pathways in CRC.
Collapse
Affiliation(s)
- Yoojeong Seo
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
- Graduate School of Medical Science, Brain Korea 21 Project, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| | - Dong Keon Kim
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| | - Jihye Park
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| | - Soo Jung Park
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| | - Jae Jun Park
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
- Yonsei Cancer Prevention Center, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| | - Jae Hee Cheon
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
- Graduate School of Medical Science, Brain Korea 21 Project, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| | - Tae Il Kim
- Division of Gastroenterology, Department of Internal Medicine, Institute of Gastroenterology, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
- Graduate School of Medical Science, Brain Korea 21 Project, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
- Yonsei Cancer Prevention Center, Severance Hospital, Yonsei University College of Medicine, Seoul 03722, Republic of Korea
| |
Collapse
|
|
4
|
Wang L, Liu H, Liu Y, Guo S, Yan Z, Chen G, Wu Q, Xu S, Zhou Q, Liu L, Peng M, Cheng X, Yan T. Potential markers of cancer stem-like cells in ESCC: a review of the current knowledge. Front Oncol 2024; 13:1324819. [PMID: 38239657 PMCID: PMC10795532 DOI: 10.3389/fonc.2023.1324819] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/20/2023] [Accepted: 12/01/2023] [Indexed: 01/22/2024] Open
Abstract
In patients with esophageal squamous cell carcinoma (ESCC), the incidence and mortality rate of ESCC in our country are also higher than those in the rest of the world. Despite advances in the treatment department method, patient survival rates have not obviously improved, which often leads to treatment obstruction and cancer repeat. ESCC has special cells called cancer stem-like cells (CSLCs) with self-renewal and differentiation ability, which reflect the development process and prognosis of cancer. In this review, we evaluated CSLCs, which are identified from the expression of cell surface markers in ESCC. By inciting EMTs to participate in tumor migration and invasion, stem cells promote tumor redifferentiation. Some factors can inhibit the migration and invasion of ESCC via the EMT-related pathway. We here summarize the research progress on the surface markers of CSLCs, EMT pathway, and the microenvironment in the process of tumor growth. Thus, these data may be more valuable for clinical applications.
Collapse
Affiliation(s)
- Lu Wang
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Huijuan Liu
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Yiqian Liu
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Shixing Guo
- Clinical Laboratory Medicine Centre, Shenzhen Hospital, Southern Medical University, Shenzhen, China
| | - Zhenpeng Yan
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Guohui Chen
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Qinglu Wu
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Songrui Xu
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Qichao Zhou
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Lili Liu
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Meilan Peng
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Xiaolong Cheng
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| | - Ting Yan
- Translational Medicine Research Center, Shanxi Medical University, Taiyuan, Shanxi, China
- Key Laboratory of Cellular Physiology of the Ministry of Education, Department of Pathology, Shanxi Medical University, Taiyuan, Shanxi, China
| |
Collapse
|
|
5
|
An L, Li M, Jia Q. Mechanisms of radiotherapy resistance and radiosensitization strategies for esophageal squamous cell carcinoma. Mol Cancer 2023; 22:140. [PMID: 37598158 PMCID: PMC10439611 DOI: 10.1186/s12943-023-01839-2] [Citation(s) in RCA: 12] [Impact Index Per Article: 12.0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/13/2023] [Accepted: 08/02/2023] [Indexed: 08/21/2023] Open
Abstract
Esophageal squamous cell carcinoma (ESCC) is the sixth most common cause of cancer-related mortality worldwide, with more than half of them occurred in China. Radiotherapy (RT) has been widely used for treating ESCC. However, radiation-induced DNA damage response (DDR) can promote the release of cytokines and chemokines, and triggers inflammatory reactions and changes in the tumor microenvironment (TME), thereby inhibiting the immune function and causing the invasion and metastasis of ESCC. Radioresistance is the major cause of disease progression and mortality in cancer, and it is associated with heterogeneity. Therefore, a better understanding of the radioresistance mechanisms may generate more reversal strategies to improve the cure rates and survival periods of ESCC patients. We mainly summarized the possible mechanisms of radioresistance in order to reveal new targets for ESCC therapy. Then we summarized and compared the current strategies to reverse radioresistance.
Collapse
Affiliation(s)
- Lingbo An
- State Key Laboratory of Cancer Biology, Department of Pathology, Xijing Hospital and School of Basic Medicine, Fourth Military Medical University, Xi'an, China
- College of Medical Technology, Xi'an Medical University, Xi'an, China
| | - Mingyang Li
- State Key Laboratory of Cancer Biology, Department of Pathology, Xijing Hospital and School of Basic Medicine, Fourth Military Medical University, Xi'an, China.
| | - Qingge Jia
- Department of Reproductive Medicine, Xi'an International Medical Center Hospital, Northwest University, Xi'an, China.
| |
Collapse
|
|
6
|
Xu K, Guo H, Xia A, Wang Z, Wang S, Wang Q. Non-coding RNAs in radiotherapy resistance: Roles and therapeutic implications in gastrointestinal cancer. Biomed Pharmacother 2023; 161:114485. [PMID: 36917887 DOI: 10.1016/j.biopha.2023.114485] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/03/2023] [Revised: 02/19/2023] [Accepted: 03/07/2023] [Indexed: 03/14/2023] Open
Abstract
Radiotherapy has become an indispensable and conventional means for patients with advanced solid tumors including gastrointestinal cancer. However, innate or acquired radiotherapy resistance remains a significant challenge and greatly limits the therapeutic effect, which results in cancer relapse and poor prognosis. Therefore, it is an urgent need to identify novel biomarkers and therapeutic targets for clarify the biological characteristics and mechanism of radiotherapy resistance. Recently, lots of studies have revealed that non-coding RNAs (ncRNAs) are the potential indicators and regulators of radiotherapy resistance via the mediation of various targets/pathways in different cancers. These findings may serve as a potential therapeutic strategy to overcome radiotherapy resistance. In this review, we will shed light on the recent findings regarding the functions and regulatory mechanisms of ncRNAs following radiotherapy, and comprehensively discuss their potential as biomarkers and therapeutic targets in radiotherapy resistance of gastrointestinal cancer.
Collapse
Affiliation(s)
- Kaiyue Xu
- Department of Hepatobiliary Surgery, The Affiliated Drum Tower Hospital of Nanjing University Medical School, Nanjing 210000, China; Department of Radiation Oncology, Suzhou Municipal Hospital, The Affiliated Suzhou Hospital of Nanjing University Medical School, Suzhou 215000, China
| | - Huimin Guo
- Department of Gastroenterology, The Affiliated Drum Tower Hospital of Nanjing University Medical School, Nanjing 210000, China
| | - Anliang Xia
- Department of Hepatobiliary Surgery, The Affiliated Drum Tower Hospital of Nanjing University Medical School, Nanjing 210000, China
| | - Zhangding Wang
- Department of Gastroenterology, The Affiliated Drum Tower Hospital of Nanjing University Medical School, Nanjing 210000, China.
| | - Shouyu Wang
- Department of Hepatobiliary Surgery, The Affiliated Drum Tower Hospital of Nanjing University Medical School, Nanjing 210000, China; Jiangsu Key Laboratory of Molecular Medicine, Nanjing University Medical School, Nanjing 210093, China.
| | - Qiang Wang
- Department of Hepatobiliary Surgery, The First Affiliated Hospital of Anhui Medical University, Hefei 230000, China; Medical Transformation Research Institute, The First Affiliated Hospital of Anhui Medical University, Hefei 230000, China.
| |
Collapse
|
|
7
|
Ergun P, Kipcak S, Bor S. Epigenetic Alterations from Barrett's Esophagus to Esophageal Adenocarcinoma. Int J Mol Sci 2023; 24:ijms24097817. [PMID: 37175524 PMCID: PMC10178512 DOI: 10.3390/ijms24097817] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 03/24/2023] [Revised: 04/17/2023] [Accepted: 04/23/2023] [Indexed: 05/15/2023] Open
Abstract
Barrett's esophagus (BE) is a disease entity that is a sequela of chronic gastroesophageal reflux disease that may result in esophageal adenocarcinoma (EAC) due to columnar epithelial dysplasia. The histological degree of dysplasia is the sole biomarker frequently utilized by clinicians. However, the cost of endoscopy and the fact that the degree of dysplasia does not progress in many patients with BE diminish the effectiveness of histological grading as a perfect biomarker. Multiple or more quantitative biomarkers are required by clinicians since early diagnosis is crucial in esophageal adenocancers, which have a high mortality rate. The presence of epigenetic factors in the early stages of this neoplastic transformation holds promise as a predictive biomarker. In this review, current studies on DNA methylations, histone modifications, and noncoding RNAs (miRNAs) that have been discovered during the progression from BE dysplasia to EAC were collated.
Collapse
Affiliation(s)
- Pelin Ergun
- Ege Reflux Study Group, Division of Gastroenterology, Faculty of Medicine, Ege University, 35040 Izmir, Türkiye
- Department of Medical Biochemistry, Faculty of Medicine, Ege University, 35040 Izmir, Türkiye
| | - Sezgi Kipcak
- Ege Reflux Study Group, Division of Gastroenterology, Faculty of Medicine, Ege University, 35040 Izmir, Türkiye
- Department of Medical Biology, Faculty of Medicine, Ege University, 35040 Izmir, Türkiye
| | - Serhat Bor
- Ege Reflux Study Group, Division of Gastroenterology, Faculty of Medicine, Ege University, 35040 Izmir, Türkiye
| |
Collapse
|
|
8
|
Noncoding RNAs in esophageal cancer: A glimpse into implications for therapy resistance. Pharmacol Res 2023; 188:106678. [PMID: 36709789 DOI: 10.1016/j.phrs.2023.106678] [Citation(s) in RCA: 8] [Impact Index Per Article: 8.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Submit a Manuscript] [Subscribe] [Scholar Register] [Received: 10/22/2022] [Revised: 01/09/2023] [Accepted: 01/25/2023] [Indexed: 01/27/2023]
Abstract
Esophageal cancer (EC) is one of the most common malignancies of the digestive system and has a high morbidity and mortality worldwide. Chemotherapy in combination with radiotherapy is one of the most important treatment modalities for EC. Chemoradiotherapy is currently acknowledged worldwide as being the standard treatment for locally advanced or unresectable disease. Unfortunately, due to the existence of therapy resistance, a number of EC patients fail to benefit from drug or irradiation treatment, which ultimately leads to poor outcomes. Considerable efforts have been made to explore the mechanisms underlying the therapy resistance of EC. Notably, noncoding RNAs (ncRNAs), including microRNAs (miRNAs), long noncoding RNAs (lncRNAs) and circular RNAs (circRNAs), are current research areas for the modulation of therapy responses and may serve as new targets to overcome treatment resistance in EC. Herein, we summarized the mechanisms by which ncRNAs are involved in drug and radiation resistance in EC and highlighted their role in promoting or repressing treatment resistance. Additionally, we discussed the clinical relevance of ncRNAs, which may serve as potential therapeutic targets and predictive biomarkers for EC.
Collapse
|
|
9
|
Wang XY, Beeraka NM, Xue NN, Yu HM, Yang Y, Liu MX, Nikolenko VN, Liu JQ, Zhao D. Identification of a three-gene prognostic signature for radioresistant esophageal squamous cell carcinoma. World J Clin Oncol 2023; 14:13-26. [PMID: 36699628 PMCID: PMC9850665 DOI: 10.5306/wjco.v14.i1.13] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Journal Information] [Submit a Manuscript] [Subscribe] [Scholar Register] [Received: 07/25/2022] [Revised: 10/25/2022] [Accepted: 12/06/2022] [Indexed: 01/10/2023] Open
Abstract
BACKGROUND Esophageal squamous cell carcinoma (ESCC) is causing a high mortality rate due to the lack of efficient early prognosis markers and suitable therapeutic regimens. The prognostic role of genes responsible for the acquisition of radioresistance in ESCC has not been fully elucidated.
AIM To establish a prognostic model by studying gene expression patterns pertinent to radioresistance in ESCC patients.
METHODS Datasets were obtained from the Gene Expression Omnibus and The Cancer Genome Atlas databases. The edgeR, a Bioconductor package, was used to analyze mRNA expression between different groups. We screened genes specifically responsible for radioresistance to estimate overall survival. Pearson correlation analysis was performed to confirm whether the expression of those genes correlated with each other. Genes contributing to radioresistance and overall survival were assessed by the multivariate Cox regression model through the calculation of βi and risk score using the following formula: 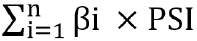 . .
RESULTS We identified three prognostic mRNAs (cathepsin S [CTSS], cluster of differentiation 180 [CD180], and SLP adapter and CSK-interacting membrane protein [SCIMP]) indicative of radioresistance. The expression of the three identified mRNAs was related to each other (r > 0.70 and P < 0.05). As to 1-year and 3-year overall survival prediction, the area under the time-dependent receiver operating characteristic curve of the signature consisting of the three mRNAs was 0.716 and 0.841, respectively. When stratifying patients based on the risk score derived from the signature, the high-risk group exhibited a higher death risk and shorter survival time than the low-risk group (P < 0.0001). Overall survival of the low-risk patients was significantly better than that of the high-risk patients (P = 0.018).
CONCLUSION We have developed a novel three-gene prognostic signature consisting of CTSS, CD180, and SCIMO for ESCC, which may facilitate the prediction of early prognosis of this malignancy.
Collapse
Affiliation(s)
- Xiao-Yan Wang
- Department of Endocrinology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Narasimha M Beeraka
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
- Department of Human Anatomy, I. M. Sechenov First Moscow State Medical University, Moscow 119991, Russia
- Department of Pharmaceutical Chemistry, JSS College of Pharmacy, Mysuru 570015, India
| | - Nan-Nan Xue
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Hui-Ming Yu
- Department of Radiation Oncology, Peking University Cancer Hospital & Institute, Beijing 065005, China
| | - Ya Yang
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Mao-Xing Liu
- Key Laboratory of Carcinogenesis and Translational Research (Ministry of Education), Department of Gastrointestinal Surgery IV, Peking University Cancer Hospital & Institute, Beijing, China
| | - Vladimir N Nikolenko
- Department of Human Anatomy, I. M. Sechenov First Moscow State Medical University, Moscow 119991, Russia
- M.V. Lomonosov Moscow State University, Moscow 119991, Russia
| | - Jun-Qi Liu
- Department of Radiation Oncology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| | - Di Zhao
- Department of Endocrinology, The First Affiliated Hospital of Zhengzhou University, Zhengzhou 450052, Henan Province, China
| |
Collapse
|
|
10
|
Chauhan N, Manojkumar A, Jaggi M, Chauhan SC, Yallapu MM. microRNA-205 in prostate cancer: Overview to clinical translation. Biochim Biophys Acta Rev Cancer 2022; 1877:188809. [PMID: 36191828 PMCID: PMC9996811 DOI: 10.1016/j.bbcan.2022.188809] [Citation(s) in RCA: 0] [Impact Index Per Article: 0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/06/2022] [Revised: 09/27/2022] [Accepted: 09/27/2022] [Indexed: 11/29/2022]
Abstract
Prostate cancer (PrCa) is the most common type of cancer among men in the United States. The metastatic and advanced PrCa develops drug resistance to current regimens which accounts for the poor management. microRNAs (miRNAs) have been well-documented for their diagnostic, prognostic, and therapeutic roles in various human cancers. Recent literature confirmed that microRNA-205 (miR-205) has been established as one of the tumor suppressors in PrCa. miR-205 regulates number of cellular functions, such as proliferation, invasion, migration/metastasis, and apoptosis. It is also evident that miR-205 can serve as a key biomarker in diagnostic, prognostic, and therapy of PrCa. Therefore, in this review, we will provide an overview of tumor suppressive role of miR-205 in PrCa. This work also outlines miR-205's specific role in targeted mechanisms for chemosensitization and radiosensitization in PrCa. A facile approach of delivery paths for successful clinical translation is documented. Together, all these studies provide a novel insight of miR-205 as an adjuvant agent for reducing the widening gaps in clinical outcome of PrCa patients.
Collapse
Affiliation(s)
- Neeraj Chauhan
- Department of Immunology and Microbiology, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA; South Texas Center of Excellence in Cancer Research, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA
| | - Anjali Manojkumar
- Department of Immunology and Microbiology, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA; South Texas Center of Excellence in Cancer Research, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA
| | - Meena Jaggi
- Department of Immunology and Microbiology, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA; South Texas Center of Excellence in Cancer Research, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA
| | - Subhash C Chauhan
- Department of Immunology and Microbiology, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA; South Texas Center of Excellence in Cancer Research, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA
| | - Murali M Yallapu
- Department of Immunology and Microbiology, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA; South Texas Center of Excellence in Cancer Research, School of Medicine, University of Texas Rio Grande Valley, McAllen, TX 78504, USA.
| |
Collapse
|
|
11
|
Qiu R, Wang W, Li J, Wang Y. Roles of PTEN inactivation and PD-1/PD-L1 activation in esophageal squamous cell carcinoma. Mol Biol Rep 2022; 49:6633-6645. [PMID: 35301651 DOI: 10.1007/s11033-022-07246-y] [Citation(s) in RCA: 4] [Impact Index Per Article: 2.0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 09/13/2021] [Revised: 02/04/2022] [Accepted: 02/08/2022] [Indexed: 02/07/2023]
Abstract
Esophageal squamous cell carcinoma (ESCC) is the most common type of esophageal cancer in China and developing countries. The purpose of this review is to summarize the roles of inactivation of the tumor suppressor gene, phosphatase and tensin homolog (PTEN), and activation of the programmed cell death protein 1 (PD-1) upon binding to its ligand (PD-L1) in the promotion of ESCC. Studies of ESCC performed in vitro and in vivo indicated that PTEN and PD-L1 function in the regulation of cell proliferation, invasion, and migration; the epithelial-mesenchymal transition; resistance to chemotherapy and radiotherapy; and the PI3K/AKT signaling pathway. Certain genetic variants of PTEN are related to susceptibility to ESCC, and PTEN and PD-L1 also function in ESCC progression and affect the prognosis of patients with ESCC. There is also evidence that the expression of PD-L1 and PTEN are associated with the progression of certain other cancers. Future studies should further examine the relationship of PD-L1 and PTEN and their possible interactions in ESCC.
Collapse
Affiliation(s)
- Rong Qiu
- Department of Radiation Oncology, Fourth Hospital of Hebei Medical University, No. 12 Jian Kang Road, Shijiazhuang, Hebei Province, P. R. China
| | - Wenxi Wang
- Department of Oncology, Xiangya Hospital, Central South University, 410008, Changsha, Hunan Province, China
| | - Juan Li
- Department of Radiation Oncology, Fourth Hospital of Hebei Medical University, No. 12 Jian Kang Road, Shijiazhuang, Hebei Province, P. R. China
| | - Yuxiang Wang
- Department of Radiation Oncology, Fourth Hospital of Hebei Medical University, No. 12 Jian Kang Road, Shijiazhuang, Hebei Province, P. R. China.
- , No.12, Jiankang Road, 050011, Shijiazhuang, Hebei Province, China.
| |
Collapse
|
|
12
|
Qiao L, Chen Y, Liang N, Xie J, Deng G, Chen F, Wang X, Liu F, Li Y, Zhang J. Targeting Epithelial-to-Mesenchymal Transition in Radioresistance: Crosslinked Mechanisms and Strategies. Front Oncol 2022; 12:775238. [PMID: 35251963 PMCID: PMC8888452 DOI: 10.3389/fonc.2022.775238] [Citation(s) in RCA: 18] [Impact Index Per Article: 9.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 09/13/2021] [Accepted: 01/24/2022] [Indexed: 12/12/2022] Open
Abstract
Radiotherapy exerts a crucial role in curing cancer, however, its treatment efficiency is mostly limited due to the presence of radioresistance. Epithelial-to-mesenchymal transition (EMT) is a biological process that endows the cancer cells with invasive and metastatic properties, as well as radioresistance. Many potential mechanisms of EMT-related radioresistance being reported have broaden our cognition, and hint us the importance of an overall understanding of the relationship between EMT and radioresistance. This review focuses on the recent progresses involved in EMT-related mechanisms in regulating radioresistance, irradiation-mediated EMT program, and the intervention strategies to increase tumor radiosensitivity, in order to improve radiotherapy efficiency and clinical outcomes of cancer patients.
Collapse
Affiliation(s)
- Lili Qiao
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Yanfei Chen
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Ning Liang
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Jian Xie
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Guodong Deng
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Fangjie Chen
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Xiaojuan Wang
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Fengjun Liu
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| | - Yupeng Li
- Department of Oncology, Shandong First Medical University, Jinan, China.,Department of General Surgery, The First Affiliated Hospital of Shandong First Medical University, Jinan, China
| | - Jiandong Zhang
- Department of Oncology, The First Affiliated Hospital of Shandong First Medical University and Shandong Province Qianfoshan Hospital, Shandong Lung Cancer Institute, Jinan, China.,Department of Oncology, Shandong First Medical University, Jinan, China
| |
Collapse
|
|
13
|
Lu Z, Yun Y, Zhang Y, Ou Y, Wang M. Promotion of microRNA-146a by histone deacetylase 4 silencing contributes to radiosensitization of esophageal carcinoma. J Transl Med 2022; 20:101. [PMID: 35193602 PMCID: PMC8862391 DOI: 10.1186/s12967-021-03171-z] [Citation(s) in RCA: 3] [Impact Index Per Article: 1.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/22/2021] [Accepted: 11/27/2021] [Indexed: 11/30/2022] Open
Abstract
Background Histone deacetylases (HDACs) have been identified to be implicated in the carcinogenesis and cancer progression. The present study was performed to probe into the effect of HDAC4 on radioresistance of esophageal carcinoma (EC). Methods The expression of HDAC4 in responders and non-responders to radiotherapy was characterized by RT-qPCR, immunohistochemistry, and Western blot analysis. EC cells were exposed to continuous fractionated X-ray irradiation, and their proliferation and apoptosis were evaluated by means of colony formation assay and flow cytometry based Annexin V-FITC/PI apoptosis assay in response to HDAC4 overexpression or silencing. Mechanistic investigation was conducted by means of in silico analysis and dual-luciferase reporter gene assay. Tumor xenografts derived from radioresistant EC cells were exposed to local X-ray irradiation in vivo for validation. Results High expression of HDAC4 was detected in either tumor tissues derived from radiotherapy responders or radioresistant EC cells. Loss of HDAC4 contributed to suppressed proliferation and enhanced apoptosis of radioresistant EC cells. Moreover, our findings revealed that HDAC4 conferred radioresistance of EC by downregulating microRNA-146a (miR-146a). Interleukin-1 receptor-associated kinase 1 (IRAK1) was a target of miR-146a, and its knockdown promoted radiosensitivity. Silencing of HDAC4 radiosensitized EC cells both in vitro and in vivo via the miR-146a/IRAK1 axis. Conclusion Hence, loss of HDAC4 upregulated miR-146a to limit radioresistance. This study aids in the better understanding about mechanism responsible for radioresistance of EC. Supplementary Information The online version contains supplementary material available at 10.1186/s12967-021-03171-z.
Collapse
Affiliation(s)
- Zhonghua Lu
- Department of Radiotherapy, Changzhou Tumor Hospital, Soochow University, Changzhou, 213000, China
| | - Yifei Yun
- Department of Radiotherapy, Changzhou Tumor Hospital, Soochow University, Changzhou, 213000, China
| | - Yutong Zhang
- Department of Radiotherapy, Changzhou Tumor Hospital, Soochow University, Changzhou, 213000, China
| | - Yao Ou
- Department of Radiotherapy, Changzhou Tumor Hospital, Soochow University, Changzhou, 213000, China
| | - Meihua Wang
- Department of Pathology, Changzhou Tumor Hospital, Soochow University, No. 68, Honghe Road, Xinbei District, Changzhou, 213000, Jiangsu, China.
| |
Collapse
|
|
14
|
Larionova I, Rakina M, Ivanyuk E, Trushchuk Y, Chernyshova A, Denisov E. Radiotherapy resistance: identifying universal biomarkers for various human cancers. J Cancer Res Clin Oncol 2022; 148:1015-1031. [PMID: 35113235 DOI: 10.1007/s00432-022-03923-4] [Citation(s) in RCA: 19] [Impact Index Per Article: 9.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 08/10/2021] [Accepted: 01/12/2022] [Indexed: 12/16/2022]
Abstract
Radiotherapy (RT) is considered as a standard in the treatment of most solid cancers, including glioblastoma, lung, breast, rectal, prostate, colorectal, cervical, esophageal, and head and neck cancers. The main challenge in RT is tumor cell radioresistance associated with a high risk of locoregional relapse and distant metastasis. Despite significant progress in understanding mechanisms of radioresistance, its prediction and overcoming remain unresolved. This review presents the state-of-the-art for the potential universal biomarkers correlated to the radioresistance and poor outcome in different cancers. We describe radioresistance biomarkers functionally attributed to DNA repair, signal transduction, hypoxia, and angiogenesis. We also focus on high throughput genetic and proteomic studies, which revealed a set of molecular biomarkers related to radioresistance. In conclusion, we discuss biomarkers which are overlapped in most several cancers.
Collapse
Affiliation(s)
- Irina Larionova
- Laboratory of Cancer Progression Biology, Cancer Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences, Tomsk, 634009, Tomsk, Russia.
| | - Militsa Rakina
- Laboratory of Translational Cellular and Molecular Biomedicine, National Research Tomsk State University, Tomsk, 634050, Tomsk, Russia
| | - Elena Ivanyuk
- Laboratory of Cancer Progression Biology, Cancer Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences, Tomsk, 634009, Tomsk, Russia
| | - Yulia Trushchuk
- Department of Gynecologic Oncology, Cancer Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences, Tomsk, 634009, Tomsk, Russia
| | - Alena Chernyshova
- Department of Gynecologic Oncology, Cancer Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences, Tomsk, 634009, Tomsk, Russia
| | - Evgeny Denisov
- Laboratory of Cancer Progression Biology, Cancer Research Institute, Tomsk National Research Medical Center, Russian Academy of Sciences, Tomsk, 634009, Tomsk, Russia
| |
Collapse
|
|
15
|
The HDAC2/SP1/miR-205 feedback loop contributes to tubular epithelial cell extracellular matrix production in diabetic kidney disease. Clin Sci (Lond) 2022; 136:223-238. [PMID: 35084460 DOI: 10.1042/cs20210470] [Citation(s) in RCA: 10] [Impact Index Per Article: 5.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/12/2021] [Revised: 01/23/2022] [Accepted: 01/27/2022] [Indexed: 11/17/2022]
Abstract
Extracellular matrix (ECM) accumulation is considered an important pathological feature of diabetic kidney disease (DKD). Histone deacetylase (HDAC) inhibitors protect against kidney injury. However, the potential mechanisms of HDACs in DKD are still largely unknown. Here, we describe a novel feedback loop composed of HDAC2 and miR-205 that regulates ECM production in tubular epithelial cells in individuals with DKD. We found that HDAC2 mRNA expression in peripheral blood was markedly higher in patients with DKD than in patients with diabetes. Nuclear HDAC2 protein expression was increased in TGFβ1-stimulated tubular epithelial cells and db/db mice. We also found that miR-205 was regulated by HDAC2 and downregulated in TGFβ1-treated HK2 cells and db/db mice. In addition, HDAC2 reduced histone H3K9 acetylation in the miR-205 promoter region to inhibit its promoter activity and subsequently suppressed miR-205 expression through an SP1-mediated pathway. Furthermore, miR-205 directly targeted HDAC2 and inhibited HDAC2 expression. Intriguingly, miR-205 also regulated its own transcription by inhibiting HDAC2 and increasing histone H3K9 acetylation in its promoter, forming a feedback regulatory loop. Additionally, the miR-205 agonist attenuated ECM production in HK2 cells and renal interstitial fibrosis in db/db mice. In conclusion, the HDAC2/SP1/miR-205 feedback loop may be crucial for the pathogenesis of DKD.
Collapse
|
|
16
|
Weiss BG, Anczykowski MZ, Ihler F, Bertlich M, Spiegel JL, Haubner F, Canis M, Küffer S, Hess J, Unger K, Kitz J, Jakob M. MicroRNA-182-5p and microRNA-205-5p as potential biomarkers for prognostic stratification of p16-positive oropharyngeal squamous cell carcinoma. Cancer Biomark 2021; 33:331-347. [PMID: 34542062 DOI: 10.3233/cbm-203149] [Citation(s) in RCA: 3] [Impact Index Per Article: 1.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 01/01/2023]
Abstract
BACKGROUND MicroRNAs constitute promising biomarkers. OBJECTIVE The aim was to investigate diagnostic and prognostic implications of miR-182-5p and miR-205-5p in p16-positive and p16-negative oropharyngeal squamous cell carcinomas (OPSCCs). METHODS Expression of miR-182-5p, miR-205-5p were determined via quantitative real-time-PCR in fresh frozen tissues of 26 p16-positive, 19 p16-negative OPSCCs and 18 HPV-negative oropharyngeal controls. Associations between miRNA-expression, clinicopathological characteristics and prognosis were analyzed. RESULTS Higher miR-182-5p expression was associated with significant inferior disease-specific survival for p16-positive OPSCCs (HR = 1.98E+09, 95% CI 0-Inf; P= 0.028) and a similar trend was observed for p16-negative OPSCCs (HR = 1.56E+09, 95% CI 0-Inf; P= 0.051). Higher miR-205-5p expression was associated with an inferior progression-free survival (HR = 4.62, 95% CI 0.98-21.83; P= 0.034) and local control rate (HR = 2.18E+09, 95% CI 0-Inf; P= 0.048) for p16-positive OPSCCs. CONCLUSIONS Results indicate that miR-182-5p and miR-205-5p can further stratify patients with p16-positive OPSCC into prognostic groups.
Collapse
Affiliation(s)
- Bernhard G Weiss
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany.,Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Mahalia Zoe Anczykowski
- Department of Otorhinolaryngology, Head and Neck Surgery, University Medical Center Göttingen, Göttingen, Germany.,Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Friedrich Ihler
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany.,German Center for Vertigo and Balance Disorders (DSGZ), Ludwig-Maximilians University of Munich, Munich, Germany
| | - Mattis Bertlich
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Jennifer L Spiegel
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Frank Haubner
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Martin Canis
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Stefan Küffer
- Institute of Pathology, University Medical Center Göttingen, Göttingen, Germany
| | - Julia Hess
- Research Unit Radiation Cytogenetics, Helmholtz Zentrum München, Research Center for Environmental Health (GmbH), Munich, Germany.,Department of Radiation Oncology, University Hospital, LMU Munich, Germany.,Clinical Cooperation Group "Personalized Radiotherapy in Head and Neck Cancer", Helmholtz Zentrum München, Research Center for Environmental Health (GmbH), Munich, Germany
| | - Kristian Unger
- Research Unit Radiation Cytogenetics, Helmholtz Zentrum München, Research Center for Environmental Health (GmbH), Munich, Germany.,Department of Radiation Oncology, University Hospital, LMU Munich, Germany.,Clinical Cooperation Group "Personalized Radiotherapy in Head and Neck Cancer", Helmholtz Zentrum München, Research Center for Environmental Health (GmbH), Munich, Germany
| | - Julia Kitz
- Institute of Pathology, University Medical Center Göttingen, Göttingen, Germany.,Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| | - Mark Jakob
- Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany.,Department of Otorhinolaryngology, Ludwig-Maximilians University of Munich, Munich, Germany
| |
Collapse
|
|
17
|
Bamps M, Dok R, Nuyts S. The DNA Damage Response Is Differentially Involved in HPV-Positive and HPV-Negative Radioresistant Head and Neck Squamous Cell Carcinoma. Cancers (Basel) 2021; 13:cancers13153717. [PMID: 34359617 PMCID: PMC8345136 DOI: 10.3390/cancers13153717] [Citation(s) in RCA: 9] [Impact Index Per Article: 3.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 06/22/2021] [Revised: 07/21/2021] [Accepted: 07/21/2021] [Indexed: 12/23/2022] Open
Abstract
Simple Summary Head and neck cancers can be divided in two major groups according to their risk factors, being high-risk human papillomavirus related (HPV-positive) and alcohol and tobacco related (HPV-negative) head and neck cancers. The majority of the locally advanced patients are treated with radiotherapy. However, up to 50% of these patients show local recurrences. The majority of these recurrences are linked to resistance to radiotherapy treatment. It is known that the response to DNA damage, also a process called the DNA damage response, is an important factor that determines the effectivity of radiotherapy. Here, we assessed the role of the DNA damage response in the resistance process to radiotherapy of head and neck cancers, by generating head and neck cancer cells resistant to radiotherapy. We show that the DNA damage response is differentially involved in the resistance process of HPV-positive and HPV-negative head and neck cancer cells. More specifically, HPV-positive radiotherapy-resistant cells showed increased ability to repair the DNA damage induced by radiotherapy. HPV-negative radiotherapy-resistant cells showed increased capacity to replicate after radiotherapy treatment. Despite this difference, inhibition of the DNA damage response enhanced the effect of radiotherapy in both groups. Abstract Radioresistance is a major cause of recurrences and radiotherapy (RT) failure in head and neck squamous cell carcinoma (HNSCC). DNA damage response (DDR) is known to be important for RT response, but its role in radioresistance is not fully understood. Here, we assessed the role of DDR in the radioresistance process of HNSCC by generating radioresistant clones from both HPV-positive SCC154 and HPV-negative SCC61 cells. We show that fractionated RT decreased RT response of HPV-positive and HPV-negative radioresistant clones in vitro and in vivo. Moreover, HPV-positive and HPV-negative radioresistant clones were characterized by differential DDR response. HPV-positive radioresistant clones showed less residual double-strand break damage and increased G2/M arrest recovery after RT, indicating an acquisition of increased DDR kinetics. In contrast, HPV-negative radioresistant clones showed less micronucleated cells after RT and increased survival upon checkpoint inhibition, indicating an increased replicative capacity. Inhibiting key factors of DDR in combination with RT rescued the radioresistant phenotype of both HPV-positive and HPV-negative radioresistant clones. Altogether, our results not only highlight the importance of DDR response in the radioresistance process of HPV-positive and HPV-negative HNSCC, but also provide possibilities for new therapies for HNSCC patients in recurrent settings.
Collapse
Affiliation(s)
- Marieke Bamps
- Laboratory of Experimental Radiotherapy, Department of Oncology, KU Leuven, University of Leuven, 3000 Leuven, Belgium
| | - Rüveyda Dok
- Laboratory of Experimental Radiotherapy, Department of Oncology, KU Leuven, University of Leuven, 3000 Leuven, Belgium
| | - Sandra Nuyts
- Laboratory of Experimental Radiotherapy, Department of Oncology, KU Leuven, University of Leuven, 3000 Leuven, Belgium
- Department of Radiation Oncology, Leuven Cancer Institute, UZ Leuven, 3000 Leuven, Belgium
| |
Collapse
|
|
18
|
Zhao Y, Jin LJ, Zhang XY. Exosomal miRNA-205 promotes breast cancer chemoresistance and tumorigenesis through E2F1. Aging (Albany NY) 2021; 13:18498-18514. [PMID: 34292880 PMCID: PMC8351670 DOI: 10.18632/aging.203298] [Citation(s) in RCA: 38] [Impact Index Per Article: 12.7] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/13/2021] [Accepted: 06/19/2021] [Indexed: 01/09/2023]
Abstract
Breast cancer (BC) is a common malignant tumor in females. The challenge in treating BC is overcoming chemoresistance. Exosome-mediated transfer of miRNAs is a molecule-shuttle in intercellular communication. Thus, we aimed to investigate whether exosomal miRNA-205 could affect chemoresistance and tumorigenesis in recipient tumor cells and to elucidate the underlying mechanism in vivo and in vitro. Microarray and qRT-PCR assays demonstrated that miRNA-205 was upregulated in tamoxifen resistance MCF-7/TAMR-1 (M/T) cells and M/T cell-derived exosomes (M/T-Exo). The M/T-Exo was internalized by human BC cells (BCCs), causing increased expression of miRNA-205 in BCCs. Coculturing with M/T-Exo promoted tamoxifen resistance, proliferation, migration, and invasion while suppressed apoptosis in recipient BCCs, which were associated with activating the caspase pathway and phosphorylating Akt. Luciferase reporter assays showed that miRNA-205 directly targeted E2F Transcription Factor 1 (E2F1) in BCCs. Furthermore, knockdown of miRNA-205 or overexpression of E2F1 reversed the roles of M/T-Exo in BCCs. In vivo experiments showed that the intratumoral injection of M/T-Exo caused greater tamoxifen resistance and larger tumor size relative to mice treated with miRNA-205-knockdown or E2F1-overexpressing BCCs. Together, the results suggest that exosomal miRNA-205 may promote tamoxifen resistance and tumorigenesis in BC through targeting E2F1 in vivo and in vitro.
Collapse
Affiliation(s)
- Yan Zhao
- Thyroid and Breast Department, Extra-Thyroid and Breast Neoplasms, Cangzhou Central Hospital, Cangzhou, Hebei, China
| | - Li-Jun Jin
- Department of Thyroid and Breast III, Cangzhou Central Hospital, Cangzhou, Hebei, China
| | - Xiao-Yu Zhang
- Department of Thyroid and Breast III, Cangzhou Central Hospital, Cangzhou, Hebei, China
| |
Collapse
|
|
19
|
Chen Y, Cui J, Gong Y, Wei S, Wei Y, Yi L. MicroRNA: a novel implication for damage and protection against ionizing radiation. ENVIRONMENTAL SCIENCE AND POLLUTION RESEARCH INTERNATIONAL 2021; 28:15584-15596. [PMID: 33533004 PMCID: PMC7854028 DOI: 10.1007/s11356-021-12509-5] [Citation(s) in RCA: 11] [Impact Index Per Article: 3.7] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Subscribe] [Scholar Register] [Received: 10/26/2020] [Accepted: 01/12/2021] [Indexed: 04/16/2023]
Abstract
Ionizing radiation (IR) is a form of high energy. It poses a serious threat to organisms, but radiotherapy is a key therapeutic strategy for various cancers. It is significant to reduce radiation injury but maximize the effect of radiotherapy. MicroRNAs (miRNAs) are posttranscriptionally regulatory factors involved in cellular radioresponse. In this review, we show how miRNAs regulate important genes on cellular response to IR-induced damage and how miRNAs participate in IR-induced carcinogenesis. Additionally, we summarize the experimental and clinical evidence for miRNA involvement in radiotherapy and discuss their potential for improvement of radiotherapy. Finally, we highlight the role that miRNAs play in accident exposure to IR or radiotherapy as predictive biomarker. miRNA therapeutics have shown great perspective in radiobiology; miRNA may become a novel strategy for damage and protection against IR.
Collapse
Affiliation(s)
- Yonglin Chen
- Hengyang Medical College, Institute of Cytology and Genetics, The Hengyang Key Laboratory of Cellular Stress Biology, University of South China, Hengyang, 421001, Hunan Province, People's Republic of China
- Hunan Province Cooperative Innovation Center for Molecular Target New Drug Study, Hengyang, 421001, Hunan Province, People's Republic of China
| | - Jian Cui
- Hengyang Medical College, Institute of Cytology and Genetics, The Hengyang Key Laboratory of Cellular Stress Biology, University of South China, Hengyang, 421001, Hunan Province, People's Republic of China
- Hunan Province Cooperative Innovation Center for Molecular Target New Drug Study, Hengyang, 421001, Hunan Province, People's Republic of China
| | - Yaqi Gong
- Hengyang Medical College, Institute of Cytology and Genetics, The Hengyang Key Laboratory of Cellular Stress Biology, University of South China, Hengyang, 421001, Hunan Province, People's Republic of China
- Hunan Province Cooperative Innovation Center for Molecular Target New Drug Study, Hengyang, 421001, Hunan Province, People's Republic of China
| | - Shuang Wei
- Hengyang Medical College, Institute of Cytology and Genetics, The Hengyang Key Laboratory of Cellular Stress Biology, University of South China, Hengyang, 421001, Hunan Province, People's Republic of China
- Hunan Province Cooperative Innovation Center for Molecular Target New Drug Study, Hengyang, 421001, Hunan Province, People's Republic of China
| | - Yuanyun Wei
- Hengyang Medical College, Institute of Cytology and Genetics, The Hengyang Key Laboratory of Cellular Stress Biology, University of South China, Hengyang, 421001, Hunan Province, People's Republic of China
- Hunan Province Cooperative Innovation Center for Molecular Target New Drug Study, Hengyang, 421001, Hunan Province, People's Republic of China
| | - Lan Yi
- Hengyang Medical College, Institute of Cytology and Genetics, The Hengyang Key Laboratory of Cellular Stress Biology, University of South China, Hengyang, 421001, Hunan Province, People's Republic of China.
- Hunan Province Cooperative Innovation Center for Molecular Target New Drug Study, Hengyang, 421001, Hunan Province, People's Republic of China.
| |
Collapse
|
|
20
|
Zhang W, Yang H, Wang Z, Wu Y, Wang J, Duan G, Guo Q, Zhang Y. miR-320a/SP1 negative reciprocal interaction contributes to cell growth and invasion in colorectal cancer. Cancer Cell Int 2021; 21:175. [PMID: 33731131 PMCID: PMC7972346 DOI: 10.1186/s12935-021-01874-3] [Citation(s) in RCA: 7] [Impact Index Per Article: 2.3] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 09/21/2020] [Accepted: 03/09/2021] [Indexed: 11/10/2022] Open
Abstract
BACKGROUND Transcription factors (TFs) may be engaged in reciprocal regulatory circuits with certain miRNAs to maintain cellular homeostasis. Disequilibrium of the reciprocities by certain tumor-related stimuli may give rise to deregulation of downstream cellular signaling pathways, thus promoting malignant tumor phenotypes. Specificity Protein 1 (SP1) is the most representative member of the tumor-related transcription factors. Previous studies disclosed that SP1 can transcriptionally regulate miRNAs and coding genes to facilitate tumor progression. In our study, we used bioinformatic analysis to predict several SP1-binding sites within the miR-320a promoter and found that SP1 is a predicted target gene of miR-320a. Therefore, we hypothesize a reciprocal regulatory link between SP1 and miR-320a that participates in colorectal cancer (CRC) development METHODS: We performed bioinformatic analysis, quantitative polymerase chain reaction (qPCR), immunoblotting, dual-luciferase reporter assays, and a series of in vitro and in vivo functional assays to describe a novel SP1/miR-320a reciprocal interaction in CRC RESULTS: First, we found that miR-320a was significantly downregulated in CRC tissues and cell lines. Consistent with findings in other cancers, miR-320a exhibited inhibitory effects on cell growth and invasion of CRC in vitro and in vivo. Moreover, we identified SP1 as a target gene of miR-320a, and ectopic SP1 expression partly abolished miR-320a-induced inhibitory effects. Conversely, we confirmed that SP1 interacts with the miR-320a promoter, leading to depression of miR-320a. This illustrates a double-negative feedback loop between miR-320a and SP1. Additionally, based on the fact that SP1 promotes MACC1 transcription, we determined via immunoblotting that the oncogenic MACC1/MET signaling pathway was inactivated in the context of miR-320a-induced SP1 downregulation CONCLUSION: Taken together, our study is the first to describe a miR-320a/SP1 negative reciprocal interaction, which contributes to cell growth and invasion in CRC through modulation of the MACC1/MET signaling pathway.
Collapse
Affiliation(s)
- Wenjing Zhang
- Department of Medical Oncology, The First People's Hospital of Yunnan Province, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, 650000, China.,Medical School, Kunming University of Science and Technology, Kunming, 650000, China
| | - Haitao Yang
- Medical School, Kunming University of Science and Technology, Kunming, 650000, China
| | - Zhongqiu Wang
- Department of Radiation Oncology, Tianjin Medical University Cancer Institute and Hospital, National Clinical Research Center for Cancer, Key Laboratory of Cancer Prevention and Therapy, Tianjin's Clinical Research Center for Cancer, Tianjin, 300060, China
| | - Yanting Wu
- Medical School, Kunming University of Science and Technology, Kunming, 650000, China
| | - Jingzhai Wang
- Department of Gastroenterology, The First People's Hospital of Yunnan Province, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, 650000, China.,Yunnan Provincial Clinical Medicine Center of Gastrointestinal Endoscopy, Kunming, 650000, China
| | - Guihua Duan
- Department of Gastroenterology, The First People's Hospital of Yunnan Province, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, 650000, China.,Yunnan Provincial Clinical Medicine Center of Gastrointestinal Endoscopy, Kunming, 650000, China
| | - Qiang Guo
- Department of Gastroenterology, The First People's Hospital of Yunnan Province, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, 650000, China. .,Yunnan Provincial Clinical Medicine Center of Gastrointestinal Endoscopy, Kunming, 650000, China.
| | - Yu Zhang
- Department of Gastroenterology, The First People's Hospital of Yunnan Province, The Affiliated Hospital of Kunming University of Science and Technology, Kunming, 650000, China. .,Yunnan Provincial Clinical Medicine Center of Gastrointestinal Endoscopy, Kunming, 650000, China.
| |
Collapse
|
|
21
|
Zhang H, Si J, Yue J, Ma S. The mechanisms and reversal strategies of tumor radioresistance in esophageal squamous cell carcinoma. J Cancer Res Clin Oncol 2021; 147:1275-1286. [PMID: 33687564 DOI: 10.1007/s00432-020-03493-3] [Citation(s) in RCA: 26] [Impact Index Per Article: 8.7] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 11/03/2020] [Accepted: 12/02/2020] [Indexed: 01/16/2023]
Abstract
Esophageal squamous cell carcinoma (ESCC) is one of most lethal malignancies with high aggressive potential in the world. Radiotherapy is used as one curative treatment modality for ESCC patients. Due to radioresistance, the 5-year survival rates of patients after radiotherapy is less than 20%. Tumor radioresistance is very complex and heterogeneous. Cancer-associated fibroblasts (CAFs), as one major component of tumor microenvironment (TME), play critical roles in regulating tumor radioresponse through multiple mechanisms and are increasingly considered as important anti-cancer targets. Cancer stemness, which renders cancer cells to be extremely resistant to conventional therapies, is involved in ESCC radioresistance due to the activation of Wnt/β-catenin, Notch, Hedgehog and Hippo (HH) pathways, or the induction of epithelial-mesenchymal transition (EMT), hypoxia and autophagy. Non-protein-coding RNAs (ncRNAs), which account for more than 90% of the genome, are involved in esophageal cancer initiation and progression through regulating the activation or inactivation of downstream signaling pathways and the expressions of target genes. Herein, we mainly reviewed the role of CAFs, cancer stemness, non-coding RNAs as well as others in the development of radioresistance and clarify the involved mechanisms. Furthermore, we summarized the potential strategies which were reported to reverse radioresistance in ESCC. Together, this review gives a systematic coverage of radioresistance mechanisms and reversal strategies and contributes to better understanding of tumor radioresistance for the exploitation of novel intervention strategies in ESCC.
Collapse
Affiliation(s)
- Hongfang Zhang
- Hangzhou Cancer Institution, Affiliated Hangzhou Cancer Hospital, Zhejiang University School of Medicine, Hangzhou, 310002, China
- Key Laboratory of Clinical Cancer Pharmacology and Toxicology Research of Zhejiang Province, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine, Hangzhou, 310006, China
| | - Jingxing Si
- Clinical Research Institute, Zhejiang Provincial People's Hospital, People's Hospital of Hangzhou Medical College, Hangzhou, 310014, China
| | - Jing Yue
- Hangzhou Cancer Institution, Affiliated Hangzhou Cancer Hospital, Zhejiang University School of Medicine, Hangzhou, 310002, China
| | - Shenglin Ma
- Hangzhou Cancer Institution, Affiliated Hangzhou Cancer Hospital, Zhejiang University School of Medicine, Hangzhou, 310002, China.
- Key Laboratory of Clinical Cancer Pharmacology and Toxicology Research of Zhejiang Province, Affiliated Hangzhou First People's Hospital, Zhejiang University School of Medicine, Hangzhou, 310006, China.
- Zhejiang University Cancer Center, Hangzhou, 310058, China.
| |
Collapse
|
|
22
|
Nguyen TQ, Hamada A, Yamada K, Higaki M, Shintani T, Yoshioka Y, Toratani S, Okamoto T. Enhanced KRT13 gene expression bestows radiation resistance in squamous cell carcinoma cells. In Vitro Cell Dev Biol Anim 2021; 57:300-314. [PMID: 33537930 DOI: 10.1007/s11626-020-00542-6] [Citation(s) in RCA: 2] [Impact Index Per Article: 0.7] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 11/23/2020] [Accepted: 12/18/2020] [Indexed: 10/22/2022]
Abstract
Cancer metastasis and recurrence are potentially lethal. A small number of cancer cell groups called cancer stem cells (CSCs) have both stem cell capacity and cancer-forming ability and are reported to play important roles in cancer metastasis and recurrence. These CSCs are considered to be radiation-resistant (RR). Therefore, understanding the biological effects of radiation on squamous cell carcinoma (SCC) cell lines in vitro and in vivo might be worthwhile to circumvent radiation resistance. Currently, there are no reports on the establishment of RR-SCC cells in serum-free defined culture, which mimics biological mechanisms and prevents instability of using serum in the culture medium. We isolated radiation-resistant strains, designated A431-LDR and A431-HDR, from A431 cells derived from vulval SCC and irradiated them with a total dose of 60 Gy at a low-dose rate (2.2 Gy/d) (RM1000) and a high-dose rate (5 Gy/5.75min) in serum-free defined culture. These cells exhibited high sphere-forming and migration ability in vitro and high tumor-forming ability in nude mice xenografts. Overexpression of KRT13 in A431-RR cells might play a role in its radiation-resistant characteristics. These cells might be useful not only to study cancer stem cells but also to study the circumvention of radiation resistance by novel cancer treatment modalities.
Collapse
Affiliation(s)
- Tam Quang Nguyen
- Department of Molecular Oral Medicine and Maxillofacial Surgery, Graduate School of Biomedical and Health Sciences, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima, 734-8553, Japan
- National Hospital of Odonto Stomatology, Ho Chi Minh City, Vietnam
| | - Atsuko Hamada
- Oral and Maxillofacial Surgery, Hiroshima University Hospital, Hiroshima, Japan
| | - Kaori Yamada
- Oral and Maxillofacial Surgery, Hiroshima University Hospital, Hiroshima, Japan
| | - Mirai Higaki
- Oral and Maxillofacial Surgery, Hiroshima University Hospital, Hiroshima, Japan
| | - Tomoaki Shintani
- Oral and Maxillofacial Surgery, Hiroshima University Hospital, Hiroshima, Japan
| | - Yukio Yoshioka
- Department of Molecular Oral Medicine and Maxillofacial Surgery, Graduate School of Biomedical and Health Sciences, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima, 734-8553, Japan
| | - Shigeaki Toratani
- Department of Molecular Oral Medicine and Maxillofacial Surgery, Graduate School of Biomedical and Health Sciences, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima, 734-8553, Japan
| | - Tetsuji Okamoto
- Department of Molecular Oral Medicine and Maxillofacial Surgery, Graduate School of Biomedical and Health Sciences, Hiroshima University, 1-2-3 Kasumi, Minami-ku, Hiroshima, 734-8553, Japan.
- School of Medical Sciences, University of East Asia, Shimonoseki 751-8503, Japan.
| |
Collapse
|
|
23
|
Huang C, Chen Y, Lai B, Chen YX, Xu CY, Liu YF. Overexpression of SP1 restores autophagy to alleviate acute renal injury induced by ischemia-reperfusion through the miR-205/PTEN/Akt pathway. JOURNAL OF INFLAMMATION-LONDON 2021; 18:7. [PMID: 33546692 PMCID: PMC7863508 DOI: 10.1186/s12950-021-00270-y] [Citation(s) in RCA: 11] [Impact Index Per Article: 3.7] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Subscribe] [Scholar Register] [Received: 11/03/2020] [Accepted: 01/21/2021] [Indexed: 12/30/2022]
Abstract
Background Acute kidney injury (AKI) is a major kidney disease with poor clinical outcome. SP1, a well-known transcription factor, plays a critical role in AKI and subsequent kidney repair through the regulation of various cell biologic processes. However, the underlying mechanism of SP1 in these pathological processes remain largely unknown. Methods An in vitro HK-2 cells with anoxia-reoxygenation injury model (In vitro simulated ischemic injury disease) and an in vivo rat renal ischemia-reperfusion injury model were used in this study. The expression levels of SP1, miR-205 and PTEN were detected by RT-qPCR, and the protein expression levels of SP1, p62, PTEN, AKT, p-AKT, LC3II, LC3I and Beclin-1 were assayed by western blot. Cell proliferation was assessed by MTT assay, and the cell apoptosis was detected by flow cytometry. The secretions of IL-6 and TNF-α were detected by ELISA. The targeted relationship between miR-205 and PTEN was confirmed by dual luciferase report assay. The expression and positioning of LC-3 were observed by immunofluorescence staining. TUNEL staining was used to detect cell apoptosis and immunohistochemical analysis was used to evaluate the expression of SP1 in renal tissue after ischemia-reperfusion injury in rats. Results The expression of PTEN was upregulated while SP1 and miR-205 were downregulated in renal ischemia-reperfusion injury. Overexpression of SP1 protected renal tubule cell against injury induced by ischemia-reperfusion via miR-205/PTEN/Akt pathway mediated autophagy. Overexpression of SP1 attenuated renal ischemia-reperfusion injury in rats. Conclusions SP1 overexpression restored autophagy to alleviate acute renal injury induced by ischemia-reperfusion through the miR-205/PTEN/Akt pathway.
Collapse
Affiliation(s)
- Chong Huang
- Department of Nephrology, The Second Affiliated Hospital of Nanchang University, 330006, Nanchang, Jiangxi Province, People's Republic of China
| | - Yan Chen
- Department of Nephrology, The Second Affiliated Hospital of Nanchang University, 330006, Nanchang, Jiangxi Province, People's Republic of China
| | - Bin Lai
- Department of Gastrointestinal Surgery, The Second Affiliated Hospital of Nanchang University, 330006, Nanchang, Jiangxi Province, People's Republic of China
| | - Yan-Xia Chen
- Department of Nephrology, The Second Affiliated Hospital of Nanchang University, 330006, Nanchang, Jiangxi Province, People's Republic of China
| | - Cheng-Yun Xu
- Department of Nephrology, The Second Affiliated Hospital of Nanchang University, 330006, Nanchang, Jiangxi Province, People's Republic of China
| | - Yuan-Fei Liu
- Department of Emergency, The Second Affiliated Hospital of Nanchang University, No.1, Minde Road, 330006, Nanchang, Jiangxi Province, People's Republic of China.
| |
Collapse
|
|
24
|
Ebahimzadeh K, Shoorei H, Mousavinejad SA, Anamag FT, Dinger ME, Taheri M, Ghafouri-Fard S. Emerging role of non-coding RNAs in response of cancer cells to radiotherapy. Pathol Res Pract 2020; 218:153327. [PMID: 33422780 DOI: 10.1016/j.prp.2020.153327] [Citation(s) in RCA: 16] [Impact Index Per Article: 4.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Submit a Manuscript] [Subscribe] [Scholar Register] [Received: 12/06/2020] [Revised: 12/23/2020] [Accepted: 12/23/2020] [Indexed: 01/03/2023]
Abstract
Radiotherapy is an effective method for treatment of a large proportion of human cancers. Yet, the efficacy of this method is precluded by the induction of radioresistance in tumor cells and the radiation-associated injury of normal cells surrounding the field of radiation. These restrictions necessitate the introduction of modalities for either radiosensitization of cancer cells or protection of normal cells against adverse effects of radiation. Non-coding RNAs (ncRNAs) have essential roles in the determination of radiosensitivity. Moreover, ncRNAs can modulate radiation-induced side effects in normal cells. Several microRNAs (miRNAs) such as miR-620, miR-21 and miR-96-5p confer radioresistance, while other miRNAs including miR-340/ 429 confer radiosensitivity. The expression levels of a number of miRNAs are associated with radiation-induced complications such as lung fibrosis or oral mucositis. The expression patterns of several long non-coding RNAs (lncRNAs) such as MALAT1, LINC00630, HOTAIR, UCA1 and TINCR are associated with response to radiotherapy. Taken together, lncRNAs and miRNAs contribute both in modulation of response of cancer cells to radiotherapy and in protection of normal cells from the associated side effects. The current review provides an overview of the roles of these transcripts in these aspects.
Collapse
Affiliation(s)
- Kaveh Ebahimzadeh
- Skull Base Research Center, Loghman Hakim Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran
| | - Hamed Shoorei
- Department of Anatomical Sciences, Faculty of Medicine, Birjand University of Medical Sciences, Birjand, Iran
| | - Seyed Ali Mousavinejad
- Skull Base Research Center, Loghman Hakim Hospital, Shahid Beheshti University of Medical Sciences, Tehran, Iran
| | | | - Marcel E Dinger
- School of Biotechnology and Biomolecular Sciences, University of New South Wales, Sydney, NSW, 2052, Australia
| | - Mohammad Taheri
- Urology and Nephrology Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
| | - Soudeh Ghafouri-Fard
- Urogenital Stem Cell Research Center, Shahid Beheshti University of Medical Sciences, Tehran, Iran.
| |
Collapse
|
|
25
|
miRNA as promising theragnostic biomarkers for predicting radioresistance in cancer: A systematic review and meta-analysis. Crit Rev Oncol Hematol 2020; 157:103183. [PMID: 33310279 DOI: 10.1016/j.critrevonc.2020.103183] [Citation(s) in RCA: 13] [Impact Index Per Article: 3.3] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/03/2020] [Revised: 11/17/2020] [Accepted: 11/23/2020] [Indexed: 02/08/2023] Open
Abstract
Radioresistance remains as an obstacle in cancer treatment. This systematic review and meta-analysis aimed to evaluate the association between the expression of miRNAs and responses to radiotherapy and the prognosis of different tumors. In total, 77 miRNAs in 19 cancer types were studied, in which 24 miRNAs were upregulated and 58 miRNAs were downregulated in cancer patients. Five miRNAs were differentially expressed. Moreover, 75 miRNAs were found to be related to radioresistance, while 5 were observed to be related to radiosensitivity. The pooled HR and 95 % confidence interval for the combined studies was 1.135 (0.819-1.574; P-value = 0.4). The HR values of the subgroup analysis for miR-21 (HR = 2.344; 95 % CI: 1.927-2.850; P-value = 0.000), nasopharyngeal carcinoma (HR = 0.448; 95 % CI: 0.265-0.760; P = 0.003) and breast cancer (HR = 1.131; 95 % CI: 0.311-4.109; P = .85) were obtained. Our results highlighted that across the published literature, miRNAs can modulate tumor radioresistance or sensitivity by affecting radiation-related signaling pathways. It seems that miRNAs could be considered as a theragnostic biomarker to predict and monitor clinical response to radiotherapy. Thus, the prediction of radioresistance in malignant patients will improve radiotherapy outcomes and radiotherapeutic resistance.
Collapse
|
|
26
|
Unveiling the ups and downs of miR-205 in physiology and cancer: transcriptional and post-transcriptional mechanisms. Cell Death Dis 2020; 11:980. [PMID: 33191398 PMCID: PMC7667162 DOI: 10.1038/s41419-020-03192-4] [Citation(s) in RCA: 31] [Impact Index Per Article: 7.8] [Reference Citation Analysis] [Abstract] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 09/10/2020] [Revised: 10/27/2020] [Accepted: 10/29/2020] [Indexed: 12/11/2022]
Abstract
miR-205 plays important roles in the physiology of epithelia by regulating a variety of pathways that govern differentiation and morphogenesis. Its aberrant expression is frequently found in human cancers, where it was reported to act either as tumor-suppressor or oncogene depending on the specific tumor context and target genes. miR-205 expression and function in different cell types or processes are the result of the complex balance among transcription, processing and stability of the microRNA. In this review, we summarize the principal mechanisms that regulate miR-205 expression at the transcriptional and post-transcriptional level, with particular focus on the transcriptional relationship with its host gene. Elucidating the mechanisms and factors regulating miR-205 expression in different biological contexts represents a fundamental step for a better understanding of the contribution of such pivotal microRNA to epithelial cell function in physiology and disease, and for the development of modulation strategies for future application in cancer therapy.
Collapse
|
|
27
|
Liu J, Xu H, Wang N, Sun M. miR-15b, a diagnostic biomarker and therapeutic target, inhibits oesophageal cancer progression by regulating the PI3K/AKT signalling pathway. Exp Ther Med 2020; 20:222. [PMID: 33363587 DOI: 10.3892/etm.2020.9352] [Citation(s) in RCA: 5] [Impact Index Per Article: 1.3] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 12/27/2019] [Accepted: 05/29/2020] [Indexed: 12/12/2022] Open
Abstract
MicroRNA (miR)-15b is an important regulator in several types of cancer, such as gastric cancer, colorectal cancer and oesophageal squamous cell carcinoma. The PI3K/AKT signalling pathway has been implicated in the growth and metastasis of oesophageal cancer (EC). The aim of the present study was to investigate the biological effects of miR-15b in EC, as well as the underlying mechanism involving the PI3K/AKT signalling pathway. The present study included 74 patients with EC and 74 healthy volunteers. The expression of miR-15b in peripheral blood mononuclear cells (PBMCs) and EC cell lines was evaluated via reverse transcription-quantitative PCR. The receiver operating characteristic curve was plotted to determine the diagnostic significance of miR-15b. EC cell viability, apoptosis, migration and invasion were analysed by conducting MTT, flow cytometry and transwell assays, respectively. Protein expression levels were analysed via western blotting. The results indicated that PBMCs isolated from patients with EC had lower miR-15b expression levels compared with PBMCs isolated from healthy volunteers. In patients with EC, miR-15b expression was strongly associated with tumour size, lymph node metastasis, TNM stage, fibrous membrane invasion and histologic grade. The results of the gain/loss-of-function in vitro experiments indicated that miR-15b inhibited EC cell viability, migration and invasion, facilitated EC cell apoptosis and attenuated the PI3K/AKT signalling pathway in EC109 and TE10 cells. Treatment of EC cells with the PI3K/AKT pathway agonist recilisib displayed the opposite effects, blocking the inhibitory function of miR-15b mimic on EC cell viability, migration and invasion. In summary, the results indicated that miR-15b suppressed EC cell viability, migration and invasion, and promoted EC cell apoptosis by inhibiting the PI3K/AKT signalling pathway.
Collapse
Affiliation(s)
- Jie Liu
- Department of Thoracic Surgery, Shandong Institute of Tumor Control, Shandong Cancer Hospital, Shandong First Medical University, Shandong Academy of Medical Sciences, Jinan, Shandong 250000, P.R. China
| | - Haiyan Xu
- The Second Department of Operating Room, Binzhou People's Hospital, Binzhou, Shandong 256610, P.R. China
| | - Nan Wang
- Laboratory Department, Affiliated Hospital of Weifang Medical College, Weifang, Shandong 261031, P.R. China
| | - Mingyan Sun
- Laboratory Department, Affiliated Hospital of Weifang Medical College, Weifang, Shandong 261031, P.R. China
| |
Collapse
|
|
28
|
Barlak N, Capik O, Sanli F, Karatas OF. The roles of microRNAs in the stemness of oral cancer cells. Oral Oncol 2020; 109:104950. [PMID: 32828020 DOI: 10.1016/j.oraloncology.2020.104950] [Citation(s) in RCA: 5] [Impact Index Per Article: 1.3] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 04/01/2020] [Revised: 07/16/2020] [Accepted: 07/28/2020] [Indexed: 02/08/2023]
Abstract
Oral cancer (OC), which is the most common form of head and neck cancers, has one of the lowest (~50%) overall 5-year survival rates. The main reasons for this high mortality rate are diagnosis of OC in advanced stages in most patients and spread to distant organs via lymph node metastasis. Many studies have shown that a small population of cells within the tumor plays vital roles in the initiation, progression, and metastasis of the tumor, resistance to chemotherapeutic agents, and recurrence. These cells, identified as cancer stem cells (CSCs), are the main reasons for the failure of current treatment modalities. Deregulated expressions of microRNAs are closely related to tumor prognosis, metastasis and drug resistance. In addition, microRNAs play important roles in regulating the functions of CSCs. Until now, the roles of microRNAs in the acquisition and maintenance of OC stemness have not been elucidated in detail yet. Here in this review, we summarized significant findings and the latest literature to better understand the involvement of CSCs in association with dysregulated microRNAs in oral carcinogenesis. Possible roles of these microRNAs in acquisition and maintenance of CSCs features during OC pathogenesis were summarized.
Collapse
Affiliation(s)
- Neslisah Barlak
- Department of Molecular Biology and Genetics, Erzurum Technical University, Erzurum, Turkey; Molecular Cancer Biology Laboratory, High Technology Application and Research Center, Erzurum Technical University, Erzurum, Turkey
| | - Ozel Capik
- Department of Molecular Biology and Genetics, Erzurum Technical University, Erzurum, Turkey; Molecular Cancer Biology Laboratory, High Technology Application and Research Center, Erzurum Technical University, Erzurum, Turkey
| | - Fatma Sanli
- Department of Molecular Biology and Genetics, Erzurum Technical University, Erzurum, Turkey; Molecular Cancer Biology Laboratory, High Technology Application and Research Center, Erzurum Technical University, Erzurum, Turkey
| | - Omer Faruk Karatas
- Department of Molecular Biology and Genetics, Erzurum Technical University, Erzurum, Turkey; Molecular Cancer Biology Laboratory, High Technology Application and Research Center, Erzurum Technical University, Erzurum, Turkey.
| |
Collapse
|
|
29
|
Mahawongkajit P, Tomtitchong P. Expression of miRNA in 5-FU resistant esophageal cancer. Mol Clin Oncol 2020; 13:221-227. [PMID: 32714549 DOI: 10.3892/mco.2020.2070] [Citation(s) in RCA: 2] [Impact Index Per Article: 0.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 12/10/2019] [Accepted: 05/04/2020] [Indexed: 02/06/2023] Open
Abstract
Fluoropyrimidine plus platinum (FP) are chemotherapeutic drugs that are most frequently used to treat esophageal squamous cell carcinoma (ESCC). However, drug resistance often occurs, and the mechanisms of resistance to 5-FU is yet to be determined. The role of micro (mi)RNAs has been well established in a variety of human cancers. The aim of the present study was to investigate the expression profile of ESCC, revealing the differential expression between ESCC and 5-FU resistant ESCC. The establishment of a 5-FU resistant (5-FUR) cell lines model provides a way of analyzing the expression of miRNAs in drug resistance. The miRNA expression indicated 50 miRNAs that were upregulated in TE10-5-FUR compared with TE10, while 119 miRNAs were downregulated. The TE11-5-FUR demonstrated 140 miRNAs were upregulated compared with TE11, which exhibited 12 downregulated miRNAs. Both cell lines share the 2 candidate upregulated miRNAs (miR-146a and miR-483-5p) and 5 downregulated miRNAs (miR-34a, miR-141, miR-200b, miR-200c and miR-205). Further studies are required to analyze and evaluate the function of the miRNAs.
Collapse
Affiliation(s)
- Prasit Mahawongkajit
- Department of Surgery, Faculty of Medicine, Thammasat University, Amphur Klongluang, Pathumthani 12120, Thailand
| | - Prakitpunthu Tomtitchong
- Department of Surgery, Faculty of Medicine, Thammasat University, Amphur Klongluang, Pathumthani 12120, Thailand
| |
Collapse
|
|
30
|
Lee TJ, Yuan X, Kerr K, Yoo JY, Kim DH, Kaur B, Eltzschig HK. Strategies to Modulate MicroRNA Functions for the Treatment of Cancer or Organ Injury. Pharmacol Rev 2020; 72:639-667. [PMID: 32554488 PMCID: PMC7300323 DOI: 10.1124/pr.119.019026] [Citation(s) in RCA: 39] [Impact Index Per Article: 9.8] [Reference Citation Analysis] [Abstract] [MESH Headings] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 12/15/2022] Open
Abstract
Cancer and organ injury-such as that occurring in the perioperative period, including acute lung injury, myocardial infarction, and acute gut injury-are among the leading causes of death in the United States and impose a significant impact on quality of life. MicroRNAs (miRNAs) have been studied extensively during the last two decades for their role as regulators of gene expression, their translational application as diagnostic markers, and their potential as therapeutic targets for disease treatment. Despite promising preclinical outcomes implicating miRNA targets in disease treatment, only a few miRNAs have reached clinical trials. This likely relates to difficulties in the delivery of miRNA drugs to their targets to achieve efficient inhibition or overexpression. Therefore, understanding how to efficiently deliver miRNAs into diseased tissues and specific cell types in patients is critical. This review summarizes current knowledge on various approaches to deliver therapeutic miRNAs or miRNA inhibitors and highlights current progress in miRNA-based disease therapy that has reached clinical trials. Based on ongoing advances in miRNA delivery, we believe that additional therapeutic approaches to modulate miRNA function will soon enter routine medical treatment of human disease, particularly for cancer or perioperative organ injury. SIGNIFICANCE STATEMENT: MicroRNAs have been studied extensively during the last two decades in cancer and organ injury, including acute lung injury, myocardial infarction, and acute gut injury, for their regulation of gene expression, application as diagnostic markers, and therapeutic potentials. In this review, we specifically emphasize the pros and cons of different delivery approaches to modulate microRNAs, as well as the most recent exciting progress in the field of therapeutic targeting of microRNAs for disease treatment in patients.
Collapse
Affiliation(s)
- Tae Jin Lee
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| | - Xiaoyi Yuan
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| | - Keith Kerr
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| | - Ji Young Yoo
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| | - Dong H Kim
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| | - Balveen Kaur
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| | - Holger K Eltzschig
- Departments of Neurosurgery (T.J.L., K.K., J.Y.Y., D.H.K., B.K.) and Anesthesiology (X.Y., H.K.E.), McGovern Medical School, University of Texas Health Science Center at Houston, Houston, Texas
| |
Collapse
|
|
31
|
Zhang KF, Wang J, Guo J, Huang YY, Huang TR. Metformin enhances radiosensitivity in hepatocellular carcinoma by inhibition of specificity protein 1 and epithelial-to-mesenchymal transition. J Cancer Res Ther 2020; 15:1603-1610. [PMID: 31939444 DOI: 10.4103/jcrt.jcrt_297_19] [Citation(s) in RCA: 3] [Impact Index Per Article: 0.8] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 11/04/2022]
Abstract
Objective Radiotherapy becomes more and more important in hepatocellular carcinoma (HCC) due to the development of technology, especially in unresectable cases. Metformin has a synergistic benefit with radiotherapy in some cancers, but remains unclear in HCC. This study aims to investigate the effect of metformin on radiosensitivity of HCC cells and the roles of specificity protein 1 (Sp1) as a target of metformin. Methods The SMMC-7721 cell line was exposed to various doses of γ-ray irradiation (0, 2, 4, 6, and 8 Gy) and with or without different concentrations of metformin (0, 1, 5, 10, and 20 mM) to measure the radiosensitivity using MTT assay. Flow cytometry was used to determine cell cycle by propidium iodide (PI) staining and apoptosis by Hoechst 33342/PI staining and Annexin V-FITC/PI staining. Real-time polymerase chain reaction and Western blotting were performed to analyze the Sp1 mRNA and protein expressions of Sp1 and epithelial-to-mesenchymal transition (EMT) marker E-cadherin and Vimentin. The invasion capability was measured by the Boyden chamber assay. Results In SMMC-7721 cells exposed to irradiation, metformin reduced proliferation and survival cells at various concentrations (0, 1, 5, 10, and 20 mM) and induced cell cycle arrest, apoptosis, and inhibited invasion. In SMMC-7721 cells with irradiation, the mRNA and protein expressions of Sp1 were significantly decreased by metformin as well as a selective Sp1 inhibitor. Metformin attenuated transforming growth factor-β1 induced decrease of E-cadherin and increase of Vimentin proteins. Conclusion Metformin demonstrated enhanced radiosensitivity and inhibition of EMT in HCC cells. Sp1 might be a target of metformin in radiosensitization.
Collapse
Affiliation(s)
- Ke-Fen Zhang
- Research Department, Affifiliated Cancer Hospital of Guangxi Medical University, Guangxi; Department of Pathology, Taishan Sanatorium, Taian, P.R. China
| | - Jun Wang
- Research Department, Affifiliated Cancer Hospital of Guangxi Medical University, Guangxi; Department of Oncology, The Central Hospital of Taian, Taian, Shandong, P.R. China
| | - Jiao Guo
- Research Department, Affifiliated Cancer Hospital of Guangxi Medical University, Guangxi, P.R. China
| | - Yue-Ying Huang
- Research Department, Affifiliated Cancer Hospital of Guangxi Medical University, Guangxi, P.R. China
| | - Tian-Ren Huang
- Research Department, Affifiliated Cancer Hospital of Guangxi Medical University, Guangxi, P.R. China
| |
Collapse
|
|
32
|
Zhou S, Zhang M, Zhou C, Wang W, Yang H, Ye W. The role of epithelial-mesenchymal transition in regulating radioresistance. Crit Rev Oncol Hematol 2020; 150:102961. [PMID: 32361589 DOI: 10.1016/j.critrevonc.2020.102961] [Citation(s) in RCA: 23] [Impact Index Per Article: 5.8] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/30/2019] [Revised: 04/06/2020] [Accepted: 04/08/2020] [Indexed: 12/16/2022] Open
Abstract
Cancer patients with different stages can benefit from radiotherapy, but there are still limited due to inherent or acquired radioresistance. The epithelial-mesenchymal transition (EMT) is a complex biological process that is implicated in malignant characteristics of cancer, such as radioresistance. Although the possible mechanisms of EMT-dependent radioresistance are being extensively studied, there is a lack of a clear picture of the overall signaling of EMT-mediated radioresistance. In this review, we highlight the role and possible molecular mechanisms of EMT in cancer radioresistance, in particular to EMT-associated signaling pathway, EMT-inducing transcription factors (EMT-TFs), EMT-related non-coding RNAs. The knowledge of EMT-associated mechanisms of radioresistance will offer more potent therapy targets to improve the radiotherapy responses.
Collapse
Affiliation(s)
- Suna Zhou
- Department of Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China; Laboratory of Cellular and Molecular Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China.
| | - Mingxin Zhang
- Department of Gastroenterology, The First Affiliated Hospital of Xi'an Medical University, Xi'an 710077, Shaanxi, China
| | - Chao Zhou
- Department of Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China; Laboratory of Cellular and Molecular Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China
| | - Wei Wang
- Department of Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China; Laboratory of Cellular and Molecular Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China
| | - Haihua Yang
- Department of Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China; Laboratory of Cellular and Molecular Radiation Oncology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China
| | - Wenguang Ye
- Department of Gastroenterology, The Affiliated Taizhou Hospital, Wenzhou Medical University, Taizhou 317000, Zhejiang, China.
| |
Collapse
|
|
33
|
Gao J, Zhang Z, Su H, Zong L, Li Y. Long Noncoding RNA FGD5-AS1 Acts as a Competing Endogenous RNA on microRNA-383 to Enhance the Malignant Characteristics of Esophageal Squamous Cell Carcinoma by Increasing SP1 Expression. Cancer Manag Res 2020; 12:2265-2278. [PMID: 32273764 PMCID: PMC7105361 DOI: 10.2147/cmar.s236576] [Citation(s) in RCA: 14] [Impact Index Per Article: 3.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/30/2019] [Accepted: 02/18/2020] [Indexed: 12/17/2022] Open
Abstract
Purpose Previous studies have identified the important roles of a long noncoding RNA called FGD5 antisense RNA 1 (FGD5-AS1) in several types of human cancer. Nonetheless, to our knowledge, the expression and functions of FGD5-AS1 in esophageal squamous cell carcinoma (ESCC) have not been clarified. In this study, we aimed to determine the expression status of long noncoding RNA FGD5-AS1 in ESCC, determine its participation in ESCC progression, and uncover the underlying mechanisms. Methods ESCC tissue samples and paired normal adjacent tissues were collected to quantify FGD5-AS1 expression by reverse-transcription quantitative PCR. The effects of FGD5-AS1 on ESCC cell proliferation, apoptosis, migration, and invasion in vitro as well as tumor growth in vivo were studied using a Cell Counting Kit-8 assay, flow cytometry, Transwell migration and invasion assays, and an in vivo tumor xenograft experiment. Results FGD5-AS1 was found to be aberrantly upregulated in both ESCC tumors and cell lines compared to the control groups. Increased FGD5-AS1 expression manifested a close association with tumor size, TNM stage, and lymph node metastasis in patients with ESCC. Overall survival of patients with ESCC was shorter in the FGD5-AS1 high-expression group than in the FGD5-AS1 low-expression group. An FGD5-AS1 knockdown markedly attenuated ESCC cell proliferation, migration, and invasion and promoted apoptosis in vitro as well as slowed tumor growth in vivo. Mechanism investigation revealed that FGD5-AS1 can increase SP1 expression by sponging microRNA-383 (miR-383), thus functioning as a competing endogenous RNA. An miR-383 knockdown and recovery of SP1 expression attenuated the inhibition of the malignant characteristics of ESCC cells by the FGD5-AS1 knockdown. Conclusion Thus, FGD5-AS1 enhances the aggressive phenotype of ESCC cells in vitro and in vivo via the miR-383–SP1 axis, which may represent a novel target for ESCC therapy.
Collapse
Affiliation(s)
- Jia Gao
- Department of Thoracic Surgery, Heze Municipal Hospital, Heze, Shandong 274031, People's Republic of China
| | - Ziteng Zhang
- Department of Thoracic Surgery, Affiliated Hospital of Jining Medical University, Shandong 272000, People's Republic of China
| | - Hong Su
- Department of Thoracic Surgery, Heze Municipal Hospital, Heze, Shandong 274031, People's Republic of China
| | - Ling Zong
- Department of Thoracic Surgery, Affiliated Hospital of Jining Medical University, Shandong 272000, People's Republic of China
| | - Yan Li
- Department of Thoracic Surgery, Heze Municipal Hospital, Heze, Shandong 274031, People's Republic of China
| |
Collapse
|
|
34
|
Wang S, Li Y, Sun S, Cai J, Cao J. Sp1 promotes ovarian cancer cell migration through repressing miR-335 expression. Biochem Biophys Res Commun 2020; 524:211-216. [PMID: 31983431 DOI: 10.1016/j.bbrc.2020.01.063] [Citation(s) in RCA: 18] [Impact Index Per Article: 4.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 12/19/2019] [Accepted: 01/11/2020] [Indexed: 12/28/2022]
Abstract
Decreased miR-335 has been reported in a variety of cancers. We previously showed that miR-335 played an important role in ovarian cancer metastasis and prognosis. However, miR-335 is down-regulated in ovarian cancer by mechanisms that remain unclear. In silico analysis identified putative transcription factor specificity protein 1 (SP1) transcription factor binding sites in the miR-335 promoter. To investigate the relation between SP1 and miR-335, qRT-PCR was performed. Our results showed both Sp1 knockdown and mithramycin A increased miR-335 expression in ovarian cancer cell lines. Luciferase reporter assays indicated that Sp1 knockdown increased miR-335 transcriptional activity. ChIP experiments showed that Sp1 bound directly to miR-335 promoter. Moreover, transwell migration and wound-healing assays showed that Sp1 knockdown resulted in inhibited cell migration, which was in turn mitigated by miR-335 inhibitor. We propose that miR-335 was negatively regulated by SP1, which in turn contributes to miR-335 deregulation and tumor cells migration.
Collapse
Affiliation(s)
- Shaohai Wang
- Department of Obstetrics and Gynecology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, China
| | - Yuan Li
- Department of Obstetrics and Gynecology, The First People's Hospital of Shangqiu, Shangqiu, China
| | - Si Sun
- Department of Obstetrics and Gynecology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, China
| | - Jing Cai
- Department of Obstetrics and Gynecology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, China
| | - Jin Cao
- Department of Obstetrics and Gynecology, Union Hospital, Tongji Medical College, Huazhong University of Science and Technology, Wuhan, 430022, China.
| |
Collapse
|
|
35
|
Gao L, Yan SB, Yang J, Kong JL, Shi K, Ma FC, Huang LZ, Luo J, Yin SY, He RQ, Hu XH, Chen G. MiR-182-5p and its target HOXA9 in non-small cell lung cancer: a clinical and in-silico exploration with the combination of RT-qPCR, miRNA-seq and miRNA-chip. BMC Med Genomics 2020; 13:3. [PMID: 31906958 PMCID: PMC6945423 DOI: 10.1186/s12920-019-0648-7] [Citation(s) in RCA: 18] [Impact Index Per Article: 4.5] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/02/2019] [Accepted: 12/19/2019] [Indexed: 12/22/2022] Open
Abstract
BACKGROUND MiR-182-5p, a cancer-related microRNA (miRNA), modulates tumorigenesis and patient outcomes in various human malignances. This study interroted the clinicopathological significance and molecular mechanisms of miR-182-5p in non-small cell lung cancer (NSCLC). METHODS The clinical significance of miR-182-5p in NSCLC subtypes was determined based on an analysis of 124 samples (lung adenocarcinomas [LUADs], n = 101; lung squamous cell carcinomas [LUSCs], n = 23) obtained from NSCLC patients and paired noncancer tissues and an analysis of data obtained from public miRNA-seq database, miRNA-chip database, and the scientific literature. The NSCLC samples (n = 124) were analyzed using the real-time quantitative polymerase chain reaction (RT-qPCR). Potential targets of miR-182-5p were identified using lists generated by miRWalk v.2.0, a comprehensive atlas of predicted and validated targets of miRNA-target interactions. Molecular events of miR-182-5p in NSCLC were unveiled based on a functional analysis of candidate targets. The association of miR-182-5p with one of the candidate target genes, homeobox A9 (HOXA9), was validated using in-house RT-qPCR and dual-luciferase reporter assays. RESULTS The results of the in-house RT-qPCR assays analysis of data obtained from public miRNA-seq databases, miRNA-chip databases, and the scientific literature all supported upregulation of the expression level of miR-182-5p level in NSCLC. Moreover, the in-house RT-qPCR data supported the influence of upregulated miR-182-5p on malignant progression of NSCLC. In total, 774 prospective targets of miR-182-5p were identified. These targets were mainly clustered in pathways associated with biological processes, such as axonogenesis, axonal development, and Ras protein signal transduction, as well as pathways involved in axonal guidance, melanogenesis, and longevity regulation, in multiple species. Correlation analysis of the in-house RT-qPCR data and dual-luciferase reporter assays confirmed that HOXA9 was a direct target of miR-182-5p in NSCLC. CONCLUSIONS The miR-182-5p expression level was upregulated in NSCLC tissues. MiR-182-5p may exert oncogenic influence on NSCLC through regulating target genes such as HOXA9.
Collapse
Affiliation(s)
- Li Gao
- Department of Pathology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Shi-Bai Yan
- Department of Medical Oncology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Jie Yang
- Department of Pharmacology, School of Pharmacy, Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Jin-Liang Kong
- Department of Respiratory and Critical Care Medicine, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Ke Shi
- Department of Pathology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Fu-Chao Ma
- Department of Medical Oncology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Lin-Zhen Huang
- Department of Pathology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Jie Luo
- Department of Medical Oncology, the Second Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Shu-Ya Yin
- Department of Pathology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Rong-Quan He
- Department of Medical Oncology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China
| | - Xiao-Hua Hu
- Department of Medical Oncology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China.
| | - Gang Chen
- Department of Pathology, the First Affiliated Hospital of Guangxi Medical University, Zhuang Autonomous Region, Nanning, 530021, Guangxi, China.
| |
Collapse
|
|
36
|
Wu H, Yu J, Kong D, Xu Y, Zhang Z, Shui J, Li Z, Luo H, Wang K. Population and single‑cell transcriptome analyses reveal diverse transcriptional changes associated with radioresistance in esophageal squamous cell carcinoma. Int J Oncol 2019; 55:1237-1248. [PMID: 31638164 PMCID: PMC6831193 DOI: 10.3892/ijo.2019.4897] [Citation(s) in RCA: 8] [Impact Index Per Article: 1.6] [Reference Citation Analysis] [Abstract] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 06/28/2019] [Accepted: 09/09/2019] [Indexed: 12/13/2022] Open
Abstract
Esophageal squamous cell carcinoma (ESCC) is a tumor composed of heterogeneous cells that easily become radioresistant, which leads to tumor recurrence. The most commonly used treatment for ESCC is fractionated irradiation (FIR) therapy that utilizes ionizing radiation to directly induce cytotoxic cell death. However, this treatment may not be able to eliminate all cancer cells due to high adaptive evolution. To determine whether the transcriptome dynamics during ESCC recurrence formation are associated with FIR response, an in vitro cell culture model for ESCC radioresistance that mimics the common radiotherapy process in patients with ESCC was established in the present study. High‑throughput sequencing analysis of in vitro cultured ESCC cells was performed using different cumulative irradiation doses, as well as tumor samples from FIR‑treated patients with ESCC before and after the development of radioresistance. Radioresistance‑associated genes and signaling pathways that were aberrantly expressed in radioresistant ESCC cells were identified, including autophagy‑related 9B (regulation of autophagy), DNA damage‑inducible transcript 4, myoglobin and plasminogen activator tissue type, which are associated with response to hypoxia, Bcl2‑binding component 3, tumor protein P63 and interferon γ‑inducible protein 16, which are associated with DNA damage response. The heterogeneity and dynamic gene expression of ESCC cells during acquired radioresistance were further studied in primary (41 single cells), 12 Gy FIR‑treated (87 single cells) and 30 Gy FIR‑treated (89 single cells) cancer cells using a single‑cell RNA sequencing approach. The results of the present study comprehensively characterized the transcriptome dynamics during acquired radioresistance in an in vitro model of ESCC and patient tumor samples at the population and single cell level. Single‑cell RNA sequencing revealed the heterogeneity of irradiated ESCC cells and an increase in the radioresistant ESCC cell subpopulation during acquired radioresistance. Overall, these results are of potential clinical relevance as they identify a number of signaling molecules associated with radioresistance, as well as opportunities for the development of novel therapeutic options for the treatment of ESCC.
Collapse
Affiliation(s)
- Hongjin Wu
- NHC Key Laboratory of Drug Addiction Medicine (Kunming Medical University), The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Juehua Yu
- NHC Key Laboratory of Drug Addiction Medicine (Kunming Medical University), The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Deshengyue Kong
- Yunnan Institute of Digestive Disease, The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Yu Xu
- NHC Key Laboratory of Drug Addiction Medicine (Kunming Medical University), The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Zunyue Zhang
- NHC Key Laboratory of Drug Addiction Medicine (Kunming Medical University), The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Jing Shui
- Shanghai International Travel Healthcare Center, Shanghai 200000, P.R. China
| | - Ziwei Li
- NHC Key Laboratory of Drug Addiction Medicine (Kunming Medical University), The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Huayou Luo
- Yunnan Institute of Digestive Disease, The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| | - Kunhua Wang
- NHC Key Laboratory of Drug Addiction Medicine (Kunming Medical University), The First Affiliated Hospital of Kunming Medical University, Kunming, Yunnan 650032, P.R. China
| |
Collapse
|
|
37
|
Yang L, Zhang X, Hou Q, Huang M, Zhang H, Jiang Z, Yue J, Wu S. Single-cell RNA-seq of esophageal squamous cell carcinoma cell line with fractionated irradiation reveals radioresistant gene expression patterns. BMC Genomics 2019; 20:611. [PMID: 31345182 PMCID: PMC6659267 DOI: 10.1186/s12864-019-5970-0] [Citation(s) in RCA: 20] [Impact Index Per Article: 4.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 11/06/2018] [Accepted: 07/11/2019] [Indexed: 01/10/2023] Open
Abstract
Background Esophageal squamous cell carcinoma (ESCC) cells are heterogeneous, easily develop radioresistance, and recur. Single-cell RNA-seq (scRNA-seq) is a next-generation sequencing method that can delineate diverse gene expression profiles of individual cells and mining their heterogeneous behaviors in response to irradiation. Our aim was using scRNA-seq to describe the difference between parental cells and cells that acquired radioresistance, and to investigate the dynamic changes of the transcriptome of cells in response to FIR. Results We sequenced ESCC cell lines KYSE180 with and without fractionated irradiation (FIR). A total of 218 scRNA-seq libraries were obtained from 88 cells exposed to 12 Gy (KYSE-180-12 Gy), 89 exposed to 30 Gy (KYSE-180-30 Gy), and 41 parental KYSE-180 cells not exposed to FIR. Dynamic gene expression patterns were determined by comprehensive consideration of genes and pathways. Biological experiments showed that KYSE-180 cells became radioresistant after FIR. PCA analysis of scRNA-seq data showed KYSE-180, KYSE-180-12 Gy and KYSE-180-30 Gy cells were discrete away from each other. Two sub-populations found in KYSE-180-12 Gy and only one remained in KYSE-180-30 Gy. This sub-population genes exposure to FIR through 12 Gy to 30 Gy were relevant to the PI3K-AKT pathway, pathways evading apoptosis, tumor cell migration, metastasis, or invasion pathways, and cell differentiation and proliferation pathways. We validated DEGs, such as CFLAR, LAMA5, ITGA6, ITGB4, and SDC4 genes, in these five pathways as radioresistant genes in bulk cell RNA-seq data from ESCC tissue of a ESCC patient treated with radiotherapy and from KYSE-150 cell lines. Conclusions Our results delineated the divergent gene expression patterns of individual ESCC cells exposure to FIR, and displayed genes and pathways related to development of radioresistance. Electronic supplementary material The online version of this article (10.1186/s12864-019-5970-0) contains supplementary material, which is available to authorized users.
Collapse
Affiliation(s)
- Ling Yang
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Xiaoyan Zhang
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Qiang Hou
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Ming Huang
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Hongfang Zhang
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Zhenzhen Jiang
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Jing Yue
- Hangzhou Cancer Institute, Hangzhou Cancer Hospital, Hangzhou, Zhejiang Province, 310002, People's Republic of China
| | - Shixiu Wu
- National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, No.113 Baohe Street Longgang District, Shenzhen, China.
| |
Collapse
|
|
38
|
Lu Y, Huang W, Chen H, Wei H, Luo A, Xia G, Deng X, Zhang G. MicroRNA-224, negatively regulated by c-jun, inhibits growth and epithelial-to-mesenchymal transition phenotype via targeting ADAM17 in oral squamous cell carcinoma. J Cell Mol Med 2019; 23:4913-4920. [PMID: 31207072 PMCID: PMC6653679 DOI: 10.1111/jcmm.14107] [Citation(s) in RCA: 12] [Impact Index Per Article: 2.4] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/27/2018] [Revised: 11/28/2018] [Accepted: 12/01/2018] [Indexed: 12/24/2022] Open
Abstract
Abnormal expression of miR-224 has been reported to promote cancer progression. However, the role of miR-224 is seldom reported in oral squamous cell carcinoma (OSCC). We reported that miR-224 expression was significantly down-regulated in OSCC tissues and cell lines. Restoration of miR-224 decreased OSCC cell growth and invasion. In addition, luciferase and Western blot assays revealed that ADAM17 protein was a downstream target of miR-224. The overexpression of ADAM17 dismissed miR-224's effect on cell growth and invasion. We concluded that miR-224 inhibited OSCC cell growth and invasion through regulating ADAM17 expression. Subsequently, we revealed that c-jun directly bind to miR-224 promoter and decreased miR-224 expression. Taken together, these findings demonstrated that miR-224 may function as a tumour-suppressive microRNA in OSCC and suggested that miR-224 may be a potential therapeutic target for OSCC patients.
Collapse
Affiliation(s)
- Yaoyong Lu
- Department of Oncology (Section 3), Gaozhou People's Hospital, Gaozhou, China
| | - Wendong Huang
- Department of Pharmacy, Maoming People's Hospital, Maoming, China
| | - Haiwen Chen
- Department of Oncology (Section 3), Gaozhou People's Hospital, Gaozhou, China
| | - Huajun Wei
- Department of Oncology (Section 3), Gaozhou People's Hospital, Gaozhou, China
| | - Aihua Luo
- Department of Pathology, Gaozhou People's Hospital, Gaozhou, China
| | - Guangsheng Xia
- Department of Otolaryngology-Head and Neck Surgery, Gaozhou People's Hospital, Gaozhou, China
| | - Xubin Deng
- Affiliated Cancer Hospital & Institute of Guangzhou Medical University, Guangzhou, China
| | - Gong Zhang
- Department of Radiotherapy, People's Hospital of Shanxi Province, Taiyuan, China
| |
Collapse
|
|
39
|
Zhang H, Jiang S, Guo L, Li X. MicroRNA-1258, regulated by c-Myb, inhibits growth and epithelial-to-mesenchymal transition phenotype via targeting SP1 in oral squamous cell carcinoma. J Cell Mol Med 2019; 23:2813-2821. [PMID: 30734471 PMCID: PMC6433684 DOI: 10.1111/jcmm.14189] [Citation(s) in RCA: 16] [Impact Index Per Article: 3.2] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/03/2018] [Revised: 01/03/2019] [Accepted: 01/10/2019] [Indexed: 01/05/2023] Open
Abstract
The biological function and underlying mechanism of miR-1258 has seldom been investigated in cancer progression, including in oral squamous cell carcinoma (OSCC). In the current study, we revealed that the expression level of miR-1258 was significantly down-regulated in OSCC tissues and cell lines. Restoration of miR-1258 decreased OSCC cell growth and invasion. The luciferase and Western blot assays revealed that SP1 protein was a downstream target of miR-1258. Overexpression of SP1 dismissed miR-1258's effect on cell growth and invasion. We also revealed that c-Myb inhibited miR-1258 by directly binding at its promoter. In addition, miR-1258 inhibited PI3K/AKT and ERK signalling pathway activity. Taken together, these findings demonstrated that miR-1258 may function as a tumour-suppressive micorRNA in OSCC and suggested that miR-1258 may be a potential therapeutic target for OSCC patients.
Collapse
Affiliation(s)
- Hua Zhang
- Department of Medical OncologyGuangdong General Hospital and Guangdong Academy of Medical SciencesGuangzhouChina
| | - Sui Jiang
- Department of Oral and Maxillofacial SurgeryGuangdong General Hospital and Guangdong Academy of Medical SciencesGuangzhouChina
| | - Longbin Guo
- Department of Operation RoomAffiliated Cancer Hospital of Guangzhou Medical UniversityGuangzhouChina
| | - Xi Li
- Department of Thyroid and Breast SurgeryThe Third Affiliated Hospital of Sun Yat‐Sen UniversityGuangzhouChina
| |
Collapse
|
|
40
|
Shao M, Li W. Transcriptional factor regulation network and competitive endogenous RNA (ceRNA) network determining response of esophageal squamous cell carcinomas to neoadjuvant chemoradiotherapy. PeerJ 2019; 7:e6668. [PMID: 30956903 PMCID: PMC6442670 DOI: 10.7717/peerj.6668] [Citation(s) in RCA: 4] [Impact Index Per Article: 0.8] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/17/2018] [Accepted: 02/21/2019] [Indexed: 01/29/2023] Open
Abstract
Background Neoadjuvant chemoradiotherapy (nCRT) followed by surgery benefits survival for patients with esophageal squamous cell carcinomas (ESCC) compared with surgery alone, but the clinical outcomes of nCRT are heterogeneous. This study aimed to elucidate transcriptional factor (TF) regulation network and competitive endogenous RNA (ceRNA) network determining response of ESCC to nCRT. Materials and Methods RNA microarray data of GSE59974 and GSE45670 were analyzed to investigate the significant changes of lincRNAs, miRNAs, mRNAs in responders and non-responders of nCRT in ESCC. Functional and enrichment analyses were conducted by clusterProfiler. The target lincRNAs and mRNAs of miRNAs were predicted by miRWalk. The ceRNA and TF regulatory networks were constructed using Cytoscape. Results Differentially expressed genes between responders and non-responders mainly enriched in biological process including Wnt signaling pathway and regulation of cell development and morphogenesis involved in differentiation. Besides, these genes showed enrichment in molecular function of glycosaminoglycan binding, metalloendopeptidase inhibitor and growth factor activity. KEGG analysis enriched these genes in pathways of neurotrophin signaling pathway, cell adhesion molecules and Wnt signaling pathway. We also constructed ceRNA network and TF network regulating response of ESCC to nCRT. Core regulatory miRNAs were miR-520a, miR-548am, miR-3184, miR-548d, miR-4725, miR-148a, miR-4659a and key regulatory TFs included MBNL1, SLC26A3, BMP4, ZIC1 and ANKRD7. Conclusion We identified significantly altered lincRNAs, miRNAs and mRNAs involved in the nCRT response of ESCC. In addition, the ceRNA regulatory network of lincRNA-miRNA-mRNA and TF regulatory network were constructed, which would elucidate novel molecular mechanisms determining nCRT response of ESCC, thus providing promising clues for clinical therapy.
Collapse
Affiliation(s)
- Mingrui Shao
- Department of Thoracic Surgery, The First Affiliated Hospital of China Medical University, Shenyang, Liaoning, China
| | - Wenya Li
- Department of Thoracic Surgery, The First Affiliated Hospital of China Medical University, Shenyang, Liaoning, China
| |
Collapse
|
|
41
|
El Bezawy R, Tinelli S, Tortoreto M, Doldi V, Zuco V, Folini M, Stucchi C, Rancati T, Valdagni R, Gandellini P, Zaffaroni N. miR-205 enhances radiation sensitivity of prostate cancer cells by impairing DNA damage repair through PKCε and ZEB1 inhibition. JOURNAL OF EXPERIMENTAL & CLINICAL CANCER RESEARCH : CR 2019; 38:51. [PMID: 30717752 PMCID: PMC6360656 DOI: 10.1186/s13046-019-1060-z] [Citation(s) in RCA: 55] [Impact Index Per Article: 11.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Subscribe] [Scholar Register] [Received: 11/21/2018] [Accepted: 01/27/2019] [Indexed: 12/15/2022]
Abstract
BACKGROUND Radiotherapy is one of the main treatment options for non-metastatic prostate cancer (PCa). Although treatment technical optimization has greatly improved local tumor control, a considerable fraction of patients still experience relapse due to the development of resistance. Radioresistance is a complex and still poorly understood phenomenon involving the deregulation of a variety of signaling pathways as a consequence of several genetic and epigenetic abnormalities. In this context, cumulative evidence supports a functional role of microRNAs in affecting radioresistance, suggesting the modulation of their expression as a novel radiosensitizing approach. Here, we investigated for the first time the ability of miR-205 to enhance the radiation response of PCa models. METHODS miR-205 reconstitution by a miRNA mimic in PCa cell lines (DU145 and PC-3) was used to elucidate miR-205 biological role. Radiation response in miRNA-reconstituted and control cells was assessed by clonogenic assay, immunofluorescence-based detection of nuclear γ-H2AX foci and comet assay. RNAi was used to silence the miRNA targets PKCε or ZEB1. In addition, target-protection experiments were carried out using a custom oligonucleotide designed to physically disrupt the pairing between the miR-205 and PKCε. For in vivo experiments, xenografts generated in SCID mice by implanting DU145 cells stably expressing miR-205 were exposed to 5-Gy single dose irradiation using an image-guided animal micro-irradiator. RESULTS miR-205 reconstitution was able to significantly enhance the radiation response of prostate cancer cell lines and xenografts through the impairment of radiation-induced DNA damage repair, as a consequence of PKCε and ZEB1 inhibition. Indeed, phenocopy experiments based on knock-down of either PKCε or ZEB1 reproduced miR-205 radiosensitizing effect, hence confirming a functional role of both targets in the process. At the molecular level, miR-205-induced suppression of PKCε counteracted radioresistance through the impairment of EGFR nuclear translocation and the consequent DNA-PK activation. Consistently, disruption of miR-205-PKCε 3'UTR pairing almost completely abrogated the radiosensitizing effect. CONCLUSIONS Our results uncovered the molecular and cellular mechanisms underlying the radiosensitizing effect of miR-205. These findings support the clinical interest in developing a novel therapeutic approach based on miR-205 reconstitution to increase PCa response to radiotherapy.
Collapse
Affiliation(s)
- Rihan El Bezawy
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Stella Tinelli
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Monica Tortoreto
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Valentina Doldi
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Valentina Zuco
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Marco Folini
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Claudio Stucchi
- Medical Physics Unit, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, 20133, Milan, Italy
| | - Tiziana Rancati
- Prostate Cancer Program, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, 20133, Milan, Italy
| | - Riccardo Valdagni
- Prostate Cancer Program, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, 20133, Milan, Italy.,Radiation Oncology 1 Unit, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, 20133, Milan, Italy
| | - Paolo Gandellini
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy
| | - Nadia Zaffaroni
- Molecular Pharmacology Unit, Department of Applied Research and Technological Development, Fondazione IRCCS Istituto Nazionale dei Tumori di Milano, Via Amadeo 42, 20133, Milan, Italy.
| |
Collapse
|
|
42
|
Qi J, Liu Y, Hu K, Zhang Y, Wu Y, Zhang X. MicroRNA-205-5p regulates extracellular matrix production in hyperplastic scars by targeting Smad2. Exp Ther Med 2019; 17:2284-2290. [PMID: 30867712 PMCID: PMC6395966 DOI: 10.3892/etm.2019.7187] [Citation(s) in RCA: 9] [Impact Index Per Article: 1.8] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/27/2017] [Accepted: 01/03/2019] [Indexed: 12/17/2022] Open
Abstract
Hypertrophic scar (HS) formation is the result of poor skin-wound healing. At present, the pathogenesis of HS formation is largely unclear. Micro (miR)RNAs have important effects on a variety of biological and pathological processes. The role of miRNA in HS formation remains largely unclear. The present study aimed to investigate the role of miR-205-5p in HS, and explore the underlying molecular mechanism. Reverse transcription-quantitative polymerase chain reaction (RT-qPCR) was used to determine the expression of miR-205-5p in HS. Western blot assay and RT-qPCR were performed to assess the expression of associated proteins and genes, respectively. TargetScan was performed to predict the target gene of miR-205-5p, and the luciferase reporter assay was applied to verify the prediction. The function of miR-205-5p on cell proliferation was detected using Cell Counting Kit-8 assay, and cell apoptosis was detected via flow cytometry. miR-205-5p expression was decreased in HS tissues and human hypertrophic scar fibroblasts (hHSFs). Mothers against decapentaplegic homolog (Smad)2 was significantly increased in HS tissues and HSFs, and it was directly targeted by miR-205-5p. Restoration of miR-205-5p suppressed HSF cell proliferation and induced cell apoptosis. It was also demonstrated that RAC-Alpha Serine/Threonine-Protein Kinase (AKT) phosphorylation and the expression of α-smooth muscle actin, collagen I and collagen III were inhibited by miR-205-5p. In addition, Smad2 weakened the effects of miR-205-5p on HSFs. In conclusion, miR-205-5p exhibited an important role in HS by targeting smad2 and suppressing the AKT pathway. These findings provide a clearer understanding of the mechanism for HS that may be used to develop novel treatments for HS.
Collapse
Affiliation(s)
- Jun Qi
- Department of Burn and Plastic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Yifei Liu
- Department of Pathology, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Kesu Hu
- Department of Burn and Plastic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Yi Zhang
- Department of Burn and Plastic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Yangyang Wu
- Department of Burn and Plastic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Xia Zhang
- Department of Burn and Plastic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| |
Collapse
|
|
43
|
Xu H, Jiang J, Zhang J, Cheng L, Pan S, Li Y. MicroRNA-375 inhibits esophageal squamous cell carcinoma proliferation through direct targeting of SP1. Exp Ther Med 2018; 17:1509-1516. [PMID: 30867685 PMCID: PMC6396021 DOI: 10.3892/etm.2018.7106] [Citation(s) in RCA: 11] [Impact Index Per Article: 1.8] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 06/11/2018] [Accepted: 09/28/2018] [Indexed: 12/20/2022] Open
Abstract
Several studies have shown that microRNA-375 (miR-375) is frequently downregulated in several types of human cancer including gastric cancer, colorectal cancer and oral squamous cell carcinoma. However, the role of miR-375 in human esophageal cancer remains unknown. In the current study, the expression level of miR-375 was analyzed in 43 esophageal squamous cell carcinoma (ESCC) tissue and matched adjacent normal tissue samples from patients with ESCC by reverse transcription-quantitative polymerase chain reaction. In addition, the expression level of miR-375 was analyzed in ESCC cell lines (KYSE450 and KYSE150) and the human esophageal epithelial cell line Het-1A by the same method. The expression level of miR-375 was significantly downregulated in ESCC tissue samples and cell lines compared with adjacent normal tissue samples and the human esophageal epithelial cell line, respectively. The effect of miR-375 on ESCC cell proliferation was detected by cell counting kit-8 (CCK-8) and colony formation assays. miR-375 overexpression significantly decreased ESCC cell proliferation and colony formation. Bioinformatics analysis was used to predict specificity protein 1 (SP1) as a target gene of miR-375 in ESCC, and this was verified by dual-luciferase assay. The present study demonstrated that SP1 regulates ESCC cell proliferation and colony formation through direct interaction with miR-375. In addition, the overall survival of patients with ESCC was analyzed using the Kaplan-Meier method and log-rank test. The results indicated that patients with ESCC with high miR-375 expression had a better survival rate compared with patients with ESCC with low miR-375 expression. Taken together, these results suggest that downregulated miR-375 promotes ESCC cell proliferation and colony formation via direct targeting of SP1, and this association may contribute to ESCC progression.
Collapse
Affiliation(s)
- Hui Xu
- Department of Anesthesiology, The First Affiliated Hospital of Anhui Medical University, Hefei, Anhui 230022, P.R. China.,Department of Anesthesiology, The Third People's Hospital of Bengbu, Bengbu, Anhui 233000, P.R. China
| | - Jialong Jiang
- Department of Anesthesiology, The First Affiliated Hospital of Anhui Medical University, Hefei, Anhui 230022, P.R. China
| | - Jingjun Zhang
- Department of Anesthesiology, The Third People's Hospital of Bengbu, Bengbu, Anhui 233000, P.R. China
| | - Liang Cheng
- Department of Anesthesiology, The Third People's Hospital of Bengbu, Bengbu, Anhui 233000, P.R. China
| | - Song Pan
- Department of Anesthesiology, The Third People's Hospital of Bengbu, Bengbu, Anhui 233000, P.R. China
| | - Yuanhai Li
- Department of Anesthesiology, The First Affiliated Hospital of Anhui Medical University, Hefei, Anhui 230022, P.R. China
| |
Collapse
|
|
44
|
Prognostic Value of MicroRNAs in Esophageal Carcinoma: A Meta-Analysis. Clin Transl Gastroenterol 2018; 9:203. [PMID: 30420592 PMCID: PMC6232177 DOI: 10.1038/s41424-018-0070-z] [Citation(s) in RCA: 18] [Impact Index Per Article: 3.0] [Reference Citation Analysis] [Abstract] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Submit a Manuscript] [Subscribe] [Scholar Register] [Received: 06/17/2018] [Revised: 09/26/2018] [Accepted: 10/08/2018] [Indexed: 12/18/2022] Open
Abstract
Background Numerous articles have reported that abnormal expression levels of microRNAs (miRNAs) are related to the survival times of esophageal carcinoma (EC) patients, which contains esophageal adenocarcinoma (EAC) and esophageal squamous cell carcinoma (ESCC). Nevertheless, there has not been a comprehensive meta-analysis to assess the accurate prognostic value of miRNAs in EC. Methods Studies published in English up to April 12, 2018 that evaluated the correlation of the expression levels of miRNAs with overall survival (OS) in EC were identified by online searches in PubMed, EMBASE, Web of Science, and the Cochrane Database of Systematic Reviews performed by two independent authors. The pooled hazard ratios (HRs) with 95% confidence intervals (CIs) were used to estimate the correlation between OS and miRNA expression. HR ≥ 2 was considered cutoff for considering the miRNA as prognostic candidate. Results Forty-four pertinent articles with 22 miRNAs and 4310 EC patients were ultimately included. EC patients with tissue expression levels of high miR-21 or low miR-133a (HR = 2.48, 95% CI = 1.50–4.12), miR-133b (HR = 2.15, 95% CI = 1.27–3.62), miR-138 (HR = 2.27, 95% CI = 1.68–3.08), miR-203 (HR = 2.83, 95% CI = 1.35–5.95), miR-375 and miR-655 (HR = 2.66, 95% CI = 1.16–6.12) had significantly poorer OS (P < 0.05). In addition, EC patients with blood expression levels of high miR-21 (HR = 2.19, 95% CI = 1.31–3.68) and miR-223 had significantly shorter OS (P < 0.05). Conclusions In conclusion, tissue expression levels of miR-21, miR-133a, miR-133b, miR-138, miR-203, miR-375, and miR-655 and blood expression levels of miR-21 and miR-223 demonstrate significant prognostic value. Among them, the expression levels of miR-133a, miR-133b, miR-138, miR-203, and miR-655 in tissue and the expression level of miR-21 in blood are potential prognostic candidates for predicting OS in EC.
Collapse
|
|
45
|
Jiang T, Zhou ML, Fan J. Inhibition of GLUT-1 expression and the PI3K/Akt pathway to enhance the chemosensitivity of laryngeal carcinoma cells in vitro. Onco Targets Ther 2018; 11:7865-7872. [PMID: 30464533 PMCID: PMC6228052 DOI: 10.2147/ott.s176818] [Citation(s) in RCA: 18] [Impact Index Per Article: 3.0] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Indexed: 12/21/2022] Open
Abstract
Background The mechanism of chemoresistance remains unknown. Here, we investigated if glucose transporter-1 (GLUT-1) and PI3K/Akt pathways are associated with the sensitivity to cisplatin in Hep-2 laryngeal carcinoma cells and whether the inhibition of GLUT-1 and the PI3K/Akt pathways enhances the chemosensitivity of Hep-2 cells. Method The effects of inhibiting GLUT-1 by a GLUT-1 siRNA, and PI3K/Akt by Ly294002, on cisplatin-induced effects were assessed in vitro. Results GLUT-1 siRNA and cisplatin showed a synergistic effect in inhibiting the proliferation of Hep-2. LY294002 and cisplatin also showed a synergistic effect in inhibiting the proliferation of Hep-2. GLUT-1 siRNA, LY294002 and cisplatin effectively inhibited the mRNA expressions and protein expressions of GLUT-1, Akt, PI3k and HIF-1α in Hep-2 cells. Furthermore, GLUT-1 siRNA and cisplatin demonstrated a synergism to inhibit the mRNA expression of HIF-1α. Moreover, it was found in this study that GLUT-1 siRNA, LY294002 and cisplatin induced the suppression of the cell cycle at G1/G2 and the increasing of apoptosis in Hep-2 cells. Conclusion This study showed that inhibiting GLUT-1, by a GLUT-1 siRNA and inhibiting PI3K/Akt by Ly294002, could suppress the proliferation of Hep-2 alone and together with cisplatin synergistically, which demonstrated the potentials to treat laryngeal carcinoma in the future therapy. Additionally, the synergistic effect between LY294002 and cisplatin to suppress the proliferation of Hep-2 might not be from GLUT-1, Akt, PI3k and HIF-1α; the synergistic effect between GLUT-1 siRNA and cisplatin to suppress the proliferation of Hep-2 might not be from GLUT-1, Akt and PI3k and might be more or less related to HIF-1α.
Collapse
Affiliation(s)
- Tao Jiang
- Department of Otolaryngology, Yinzhou People's Hospital of Ningbo City Zhejiang Province, Zhejiang, China,
| | | | - Jun Fan
- State Key Laboratory for Diagnosis and Treatment of Infectious Diseases, The First Affiliated Hospital, College of Medicine Zhejiang University, Zhejiang, China
| |
Collapse
|
|
46
|
Malhotra A, Sharma U, Puhan S, Chandra Bandari N, Kharb A, Arifa PP, Thakur L, Prakash H, Vasquez KM, Jain A. Stabilization of miRNAs in esophageal cancer contributes to radioresistance and limits efficacy of therapy. Biochimie 2018; 156:148-157. [PMID: 30326253 DOI: 10.1016/j.biochi.2018.10.006] [Citation(s) in RCA: 30] [Impact Index Per Article: 5.0] [Reference Citation Analysis] [Abstract] [Key Words] [MESH Headings] [Grants] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 05/05/2018] [Accepted: 10/11/2018] [Indexed: 12/15/2022]
Abstract
The five-year survival rate of esophageal cancer patients is less than 20%. This may be due to increased resistance (acquired or intrinsic) of tumor cells to chemo/radiotherapies, often caused by aberrant cell cycle, deregulated apoptosis, increases in growth factor signaling pathways, and/or changes in the proteome network. In addition, deregulation in non-coding RNA-mediated signaling pathways may contribute to resistance to therapies. At the molecular level, these resistance factors have now been linked to various microRNA (miRNAs), which have recently been shown to control cell development, differentiation and neoplasia. The increased stability and dysregulated expression of miRNAs have been associated with increased resistance to various therapies in several cancers, including esophageal cancer. Therefore, miRNAs represent the next generation of molecules with tremendous potential as biomarkers and therapeutic targets. However, detailed studies on miRNA-based therapeutic interventions are still in their infancy. Hence, in this review, we have summarized the current status of microRNAs in dictating the resistance/sensitivity of tumor cells to chemotherapy and radiotherapy. In addition, we have discussed various strategies to increase radiosensitivity, including targeted therapy, and the use of miRNAs as radiosensitive/radioresistance biomarkers for esophageal cancer in the clinical setting.
Collapse
Affiliation(s)
- Akshay Malhotra
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - Uttam Sharma
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - Shyamly Puhan
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - Naga Chandra Bandari
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - Anjali Kharb
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - P P Arifa
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - Lovlesh Thakur
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India
| | - Hridayesh Prakash
- Laboratory Oncology Unit, Institute Rotary Cancer Hospital, All India Institute of Medical Sciences, Ansari Nagar, New Delhi, 110029, India; Institute of Virology and Immunology, Amity University, NOIDA, India.
| | - Karen M Vasquez
- Division of Pharmacology and Toxicology, College of Pharmacy, The University of Texas at Austin, Dell Pediatric Research Institute, 1400 Barbara Jordan Blvd, Austin, TX, 78723, USA
| | - Aklank Jain
- Department of Animal Sciences, Central University of Punjab, Bathinda, Punjab, India.
| |
Collapse
|
|
47
|
miR-497/Wnt3a/c-jun feedback loop regulates growth and epithelial-to-mesenchymal transition phenotype in glioma cells. Int J Biol Macromol 2018; 120:985-991. [PMID: 30171955 DOI: 10.1016/j.ijbiomac.2018.08.176] [Citation(s) in RCA: 14] [Impact Index Per Article: 2.3] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 02/04/2018] [Revised: 08/27/2018] [Accepted: 08/28/2018] [Indexed: 12/31/2022]
Abstract
Glioma is one of the most frequent intracranial malignant tumors. Abnormal expression of microRNAs usually contributes to the development and progression of glioma. In the current study, we explored the role and underlying mechanism of miR-497 in glioma. We revealed that miR-497 expression was significantly down-regulated in glioma tissues and cell lines. Reduced expression of miR-497 was associated with poor disease-free and over-all survival rate. Restoration of miR-497 decreased glioma cell growth and invasion both in vitro and in vivo. The oncogene Wnt3a was identified as a downstream target of miR-497 by using luciferase and western blot assays. Knockdown of Wnt3a mimicked the effect of miR-497 in glioma cells. In summary, our study demonstrated that miR-497 may function as a tumor suppressor in glioma and suggested that miR-497 is a potential therapeutic target for glioma patients.
Collapse
|
|
48
|
Shi H, Mao Y, Ju Q, Wu Y, Bai W, Wang P, Zhang Y, Jiang M. C-terminal binding protein‑2 mediates cisplatin chemoresistance in esophageal cancer cells via the inhibition of apoptosis. Int J Oncol 2018; 53:167-176. [PMID: 29658564 DOI: 10.3892/ijo.2018.4367] [Citation(s) in RCA: 9] [Impact Index Per Article: 1.5] [Reference Citation Analysis] [Abstract] [Track Full Text] [Journal Information] [Subscribe] [Scholar Register] [Received: 10/18/2017] [Accepted: 03/09/2018] [Indexed: 11/05/2022] Open
Abstract
C-terminal binding protein‑2 (CtBP2) is a transcriptional co-repressor that is associated with tumorigenesis and tumor progression. It has been reported to predict a poor prognosis in several human cancers, including esophageal squamous cell carcinoma (ESCC). The present study aimed to investigate the involvement of CtBP2 in the cisplatin (DDP) resistance of the ECA109 ESCC cell line and its effect on the expression of apoptosis-associated proteins. Constructed recombinant lentiviruses were used for the knockdown or overexpression of CtBP2 in ECA109 cells, and the expression of CtBP2 was measured using reverse transcription-quantitative polymerase chain reaction and western blotting following transfection. MTT assays, Hoechst 33342 staining and flow cytometry (FCM) were applied to detect the influence of CtBP2 on the DDP-induced viability and apoptosis of the transfected ECA109 cells. In addition, the levels of apoptosis-associated proteins, including p53, B‑cell lymphoma 2 (Bcl‑2), Bcl‑2‑associated X protein (Bax) and activated caspase-3 were investigated in the transfected ECA109 cells. Stable ECA109 cells with CtBP2 overexpression or knockdown were successfully established. The results of the MTT, Hoechst 33342 and FCM assays demonstrated that overexpression of CtBP2 attenuated the reduction of cell viability and inhibited the cell apoptosis induced by DDP. Furthermore, the western blotting results indicated that CtBP2 overexpression inhibited the DDP-induced apoptosis of ECA109 cells via the reduction of p53, activated caspase-3 and Bax expression, and promotion of Bcl‑2 expression. Therefore, the present study indicated that CtBP2 reduced the susceptibility of ECA109 cells to DDP by regulating the expression of apoptosis-related proteins, suggesting that it may be a promising therapeutic target in ESCC in the future.
Collapse
Affiliation(s)
- Hui Shi
- Laboratory Animals Center, Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Yinting Mao
- Department of Thoracic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Qianqian Ju
- Department of Thoracic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Yingcheng Wu
- Medical College of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Wen Bai
- Medical College of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Peiwen Wang
- Medical College of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Yudong Zhang
- Department of Thoracic Surgery, Affiliated Hospital of Nantong University, Nantong, Jiangsu 226001, P.R. China
| | - Maorong Jiang
- Laboratory Animals Center, Nantong University, Nantong, Jiangsu 226001, P.R. China
| |
Collapse
|
|
49
|
Chu P, Liang A, Jiang A, Zong L. miR-205 regulates the proliferation and invasion of ovarian cancer cells via suppressing PTEN/SMAD4 expression. Oncol Lett 2018; 15:7571-7578. [PMID: 29725462 PMCID: PMC5920363 DOI: 10.3892/ol.2018.8313] [Citation(s) in RCA: 29] [Impact Index Per Article: 4.8] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 01/04/2017] [Accepted: 11/16/2017] [Indexed: 12/21/2022] Open
Abstract
MicroRNAs (miRNAs/miRs) are non-coding RNAs that post-transcriptionally control target genes, and are involved in tumorigenesis, apoptosis, proliferation, invasion, metastasis and chemoresistance. However, data concerning miRNAs in ovarian cancer remain incomplete. The present study aimed to identify miRNAs that affected the malignant phenotype of ovarian cancer, and to analyze their potential mechanisms. The data demonstrated that miR-205 promoted cell proliferation and invasion of ovarian cancer cells via suppressing Phosphatase and tensin homolog (PTEN)/mothers against decapentaplegic homolog 4 (SMAD4) expression. Based on the Cancer Genome Atlas database analysis results, it was identified that miR-205 was significantly upregulated in ovarian cancer tissues and markedly correlated with poor prognosis in patients with ovarian cancer; its abnormal expression was also confirmed in tissues from patients with ovarian cancer by reverse transcription quantitative polymerase chain reaction. Additional Gene Ontology analysis revealed that the target genes of miR-205 were associated with cell proliferation and invasion. Consistent with the database analysis, miR-205 overexpression significantly promoted ovarian cancer cell proliferation and invasion in vitro. To additionally explore the mechanism by which miR-205 was associated with proliferation and invasion of ovarian cancer cells, a protein-protein interaction network was constructed based on miR-205 target genes associated with proliferation and invasion, and it was revealed that PTEN and SMAD4 were key target genes of miR-205. In ovarian cancer tissues, the expression levels of PTEN and SMAD4 were significantly downregulated, suggesting that miR-205 may suppress the expression of PTEN and SMAD4 in vivo. In vitro, miR-205 overexpression markedly suppressed the expression of SMAD4 and PTEN, additionally verifying that PTEN and SMAD4 were the target genes of miR-205 in ovarian cancer cells. These results elucidated the tumor-promoting role of miR-205 and established miR-205 as a potential treatment target for ovarian cancer.
Collapse
Affiliation(s)
- Ping Chu
- Department of Gynecology, Yantai Affiliated Hospital of Binzhou Medical University, Yantai, Shandong 264100, P.R. China
| | - Aihua Liang
- Department of Gynecology, Yantai Affiliated Hospital of Binzhou Medical University, Yantai, Shandong 264100, P.R. China
| | - Aili Jiang
- Department of Gynecology, Yantai Affiliated Hospital of Binzhou Medical University, Yantai, Shandong 264100, P.R. China
| | - Lu Zong
- Department of Gynecology and Obstetrics, The First Affiliated Hospital of Xi'an Jiaotong University, Xi'an, Shaanxi 710061, P.R. China
| |
Collapse
|
|
50
|
Di Agostino S, Valenti F, Sacconi A, Fontemaggi G, Pallocca M, Pulito C, Ganci F, Muti P, Strano S, Blandino G. Long Non-coding MIR205HG Depletes Hsa-miR-590-3p Leading to Unrestrained Proliferation in Head and Neck Squamous Cell Carcinoma. Theranostics 2018; 8:1850-1868. [PMID: 29556360 PMCID: PMC5858504 DOI: 10.7150/thno.22167] [Citation(s) in RCA: 52] [Impact Index Per Article: 8.7] [Reference Citation Analysis] [Abstract] [Key Words] [Track Full Text] [Download PDF] [Figures] [Journal Information] [Subscribe] [Scholar Register] [Received: 07/31/2017] [Accepted: 12/24/2017] [Indexed: 12/30/2022] Open
Abstract
Over 70% of head & neck squamous cell carcinoma (HNSCC) patients carry TP53 oncogenic mutations. Here we studied the role of specific tumor-derived mutant p53 proteins in the aberrant transcription of long non-coding (lnc) MIR205HG gene in head and neck cancer cells. Methods: To understand the role of lncMIR205HG, that we showed to be transcriptionally regulated by mutant p53 in HNSCC, we have employed siRNA and shRNA in CAL27 and FaDu HNSCC cell lines to suppress p53 gene expression in ChIP assays and RT-qPCR. We validated our findings in a cohort of 522 HNSCC patients from The Cancer Genome Atlas Data Portal (TCGA). We further evaluated our results in 63 HNSCC tumor samples collected at our institute, 32 of which were characterized by mutated TP53 (missense mutations) while 31 were characterized by wild-type TP53. Results: Maturation of pre-MIR205HG transcript produces two non-coding RNAs, lncMIR205HG and hsa-miR-205-5p. Down-regulation of lncMIR205HG expression significantly reduced cell proliferation, cell migration and clonogenic activity of head and neck cancer cells. Expression of MIR205HG was significantly increased in HNSCC with mutated TP53 when compared with matched non-tumoral tissues. Furthermore, MIR205HG expression levels were significantly higher in tumoral samples with mutant p53 than in tumoral tissues expressing wild-type p53. Mechanistically, MIR205HG depletes endogenous miR-590-3p leading to increased cyclin B, cdk1, and YAP protein expression. Conclusions: Taken together, these findings identify a transcriptional and post-transcriptional molecular network that includes mutant p53 protein, lncMIR205HG, YAP, and other proliferation-related genes, which are enriched in HNSCC patients with poor prognosis.
Collapse
|